In the 19th century, the research on the structure of the atom was yielding incomprehensible results. Scientists worldwide were astounded by the revelations. However, this situation also increased the desire to discover more about the atom. It sparked interest in understanding the nature of the universe.
Discovery of the Neutron
It is remarkable that during this research, the neutron was not discovered. It is a very fundamental particle of the atom. In 1920, Ernest Rutherford pointed out the disparity between the atomic number and the atomic mass of atoms. However, it was in 1932 that people came to know about this particle.
The discovery of neutron started with some unanswered questions. Why do many elements have mass more than the mass of their protons? Helium atom was the simplest example. Why does Helium (Z=2) have mass four times that of hydrogen (Z=1)? It was known that electrons don’t have significant mass. So all the mass of an atom is actually the mass of its protons. If Helium has 2 protons and hydrogen has 1, helium’s mass should ideally be double that of hydrogen. However, the helium atom is four times heavier than hydrogen. This observation could not be explained.
Mass of helium = 4× (Mass of hydrogen)…WHY?
Ernest Rutherford concluded that another particle might exist in the nucleus. This particle is electrically neutral. However, there was no solid evidence to prove that.

In 1932, that James Chadwick, an English Physicist, discovered this sub-atomic particle!
In his undergraduate days (1911), James Chadwick was Rutherford’s student at Victoria University of Manchester. In 1913, he was awarded a research scholarship. He went to Berlin to study under Hans Geiger. The first world war broke out in 1914. James Chadwick became a prisoner of war in Germany. When the war ended, after 4 years, he went back to his mentor, Ernest Rutherford in Cambridge University. In 1921, Rutherford made him assistant lab director at the Cavendish lab at Cambridge University.
Rutherford assigned Chadwick the task of finding out the neutral particle, hidden in the nucleus, that he had predicted earlier. The discovery of this particle started with some experiments conducted in different labs.
In 1931, Walther Bothe and Herbert Becker made an important discovery. They found that α-particles emitted strong radiation when they hit a target of a lighter metal like Beryllium. The same was true for metals like Lithium and Boron. This radiation was thought to be the gamma rays. However, later it became evident that it was something else. This radiation was far more penetrating than the gamma radiation!
Irene Curie and her husband discovered that when a beam of this radiation hit a substance rich in protons, e.g.– paraffin (an alkane), protons were knocked off from it. These protons could be detected by a Geiger counter.
The James Chadwick Experiment
James Chadwick started his work based on the observations of the above experiments. He started conducting experiments to find out the nature of this radiation. The experiment that gave him the desired results was as follows-

He used Polonium as a source of α- particles. Polonium (a radioactive element) emits α- particles, which then hit the beryllium foil. This lead to the emission of some kind of radiation. This emitted radiation was not deflected by an electric field. Thus, he concluded that it had to be electrically neutral. The radiation was very penetrating. Thus, it could knock off the protons (H+) from the paraffin slab. As soon as these protons (H+) entered the gas-filled chamber, they ionised the gas within. The recorder, a Geiger counter, recorded the amount of gas ionized. The amount of gas ionized was proportional to the number of protons entering the chamber.
James Chadwick proposed that the neutral particle in the Beryllium atom’s nucleus caused this effect on the paraffin. He concluded that this was the very particle which Rutherford had predicted earlier. Thus, he discovered the neutron! The nuclear reaction that produced neutrons was as follows –
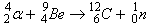
James Chadwick not only discovered the neutron but also found the mass of this neutron. He observed that the mass of a neutron was comparable to the proton’s mass. It is slightly larger than the proton mass. In 1932, he was awarded the noble prize for this discovery!
Thus, three fundamental particles of the atom were discovered – THE PROTON, THE ELECTRON, and THE NEUTRON.
It could now be fully understood, why the mass of the Helium atom was 4 times the mass of the hydrogen atom. The helium atom consists of 2 protons and 2 neutrons, which accounted for its mass. The electrons revolve around the nucleus as shown below-
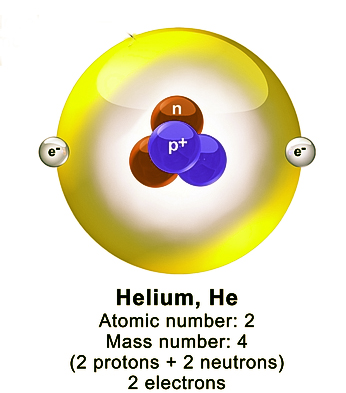
So, was this the final structure of the atom? Was this an inerrant model? Let us find the answers to this question in the next post. Till then,
Be a perpetual student of life and keep learning…
Good Day!
References and further reading –
1) https://en.wikipedia.org/wiki/Discovery_of_the_neutron
2)https://sites.google.com/site/chadwickexperiment/home
3)http://www-outreach.phy.cam.ac.uk/camphy/neutron/neutron5_1.htm
4)http://www.physicslab.org/Document.aspx?doctype=3&filename=AtomicNuclear_ChadwickNeutron.xml
Image source –
1)https://en.wikipedia.org/wiki/File:James_Chadwick.jpg
2)http://www.physicslab.org/Document.aspx?doctype=3&filename=AtomicNuclear_ChadwickNeutron.xml
3)By BruceBlaus – Own work, CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=33041232