The Valence bond theory (VBT) is one of the theories which explains the formation of the covalent bond. It is based on the assumption that atomic orbitals overlap and form a bond. The electrons are shared between the two atoms and the bond represents the region where the overlap occurs – the region with maximum electron density. Let us now discuss the various types of overlap and the types of bonds formed.
- Pi (Π) bond – This bond is formed when the overlapping takes place perpendicular to the internuclear distance /bond axis. Electron density is in two lobes (see ‘region of overlap’ in the figure below). There is one nodal plane (2 nodes – one each of one p orbital). Please note that, although there are two regions of overlap, it represents only ONE Π bond.
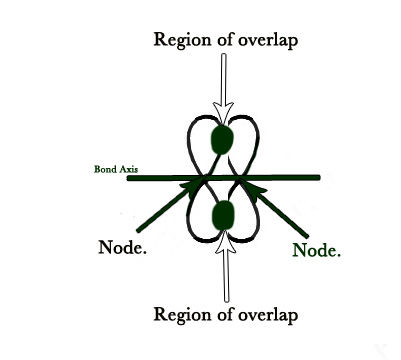
Double and triple bonded molecules have 1 and 2 pi bonds respectively.A pi bond is always formed only in addition to a sigma bond. It is because the atoms constituting a single bond prefer to form a strong sigma bond rather than a weak pi bond. Thus, a pi bond is always present in molecules having multiple bonds, i.e., double or triple bonds. In other words, a single bond cannot be a pi bond.
Single bond → σ bond
Double bond → 1 σ bond + 1 Π bond.
Triple bond → 1 σ bond + 2 Π bonds.
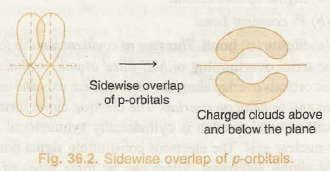
The electron charge cloud of the Π-bond is placed above and below the plane of bonding atoms. This results in the electrons being readily available to the attacking reagents as these electrons are not directly in the path of the nucleus and are so loosely held. Thus, molecules with Π -bonds are more reactive.
e.g. – Alkenes and alkynes are more reactive than alkanes.
Sigma bond | Pi bond |
| overlap along the bond axis | overlap perpendicular to the bond axis |
| overlap is greater in a sigma bond, so it is a strong bond | overlap(sideways ) is lesser compared to the sigma bond, thus the bond is weaker. |
| s- and p- orbitals in the plane of the inter-nuclear axis form a sigma bond | only the unhybridized p-orbitals perpendicular to the plane of the molecule form pi bonds. |
TYPES OF OVERLAP OF ATOMIC ORBITALS FORMING A SIGMA BOND –
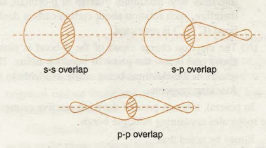
There are three different types of σ overlaps that we encounter generally. These overlaps are between the s and p- orbitals. The three different overlaps are as follows –
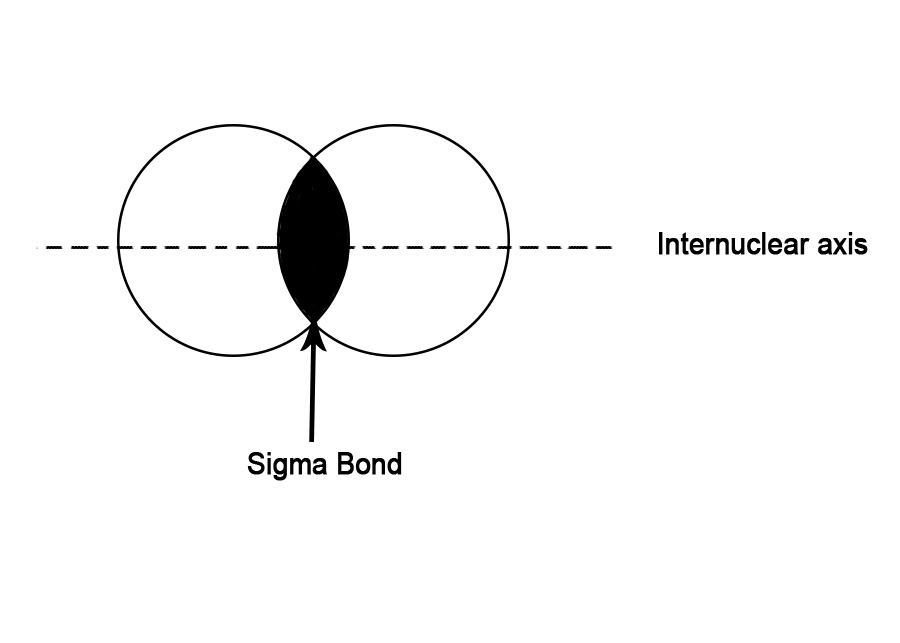
i) s-s overlap – The overlap occurs between two half-filled s-orbitals of two adjacent atoms. As s- orbitals are spherically symmetrical, the overlap always takes place axially i.e along the bond axis. Thus, this is a σ bond.
e.g.- Formation of H2 molecule –
Electronic Configuration of H(1) → 1s1.
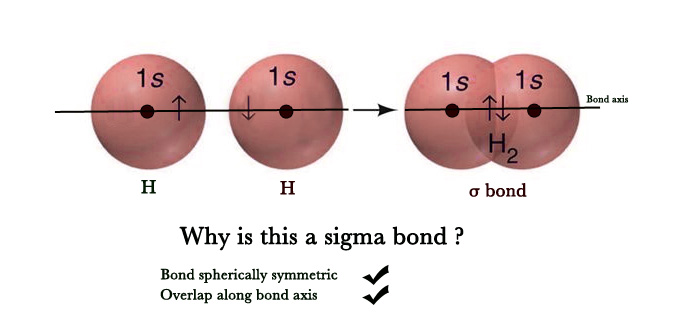
In the above figure, s- orbitals of two hydrogen atoms overlap axially to form a sigma bond.
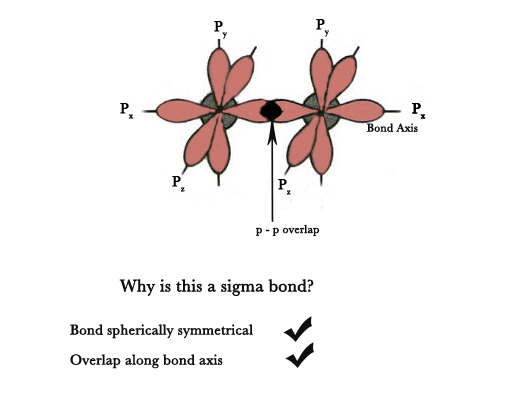
ii) p-p overlap – The overlap occurs between two half-filled p-orbitals of two adjacent atoms containing electrons with opposite spins( p- orbitals are dumb bell shaped -not spherically symmetrical). However, as the overlap shown in the figure below takes place axially i.e along the bond axis, the bond formed is a σ bond.
e.g.- Formation of F2 molecule –
Electronic Configuration of F(9) → 1s1 2s2 2px1 2py2 2pz2 .
As shown below, the two half-filled px orbitals of two fluorine atoms overlap along the bond axis, to form a sigma covalent bond. The electron density lies between the two nuclei. Thus, F2 molecule is formed.
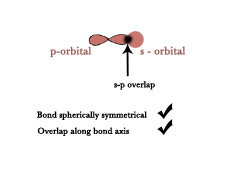
iii) s-p overlap – This overlap takes place between the s-orbital of one atom and the p- orbital of the other atom along the bond axis.
e.g.- Formation of HF molecule –
Electronic Configuration of H(1) → 1s1.
Electronic Configuration of F(9) → 1s1 2s2 2px1 2py2 2pz2 .
As shown below, one half-filled s orbital of hydrogen overlaps with a half-filled p-orbital of fluorine atom along the bond axis.
Note – when two p orbitals overlap along the bond axis sigma bond is formed and when they overlap perpendicular to the bond axis a pi bond is formed.
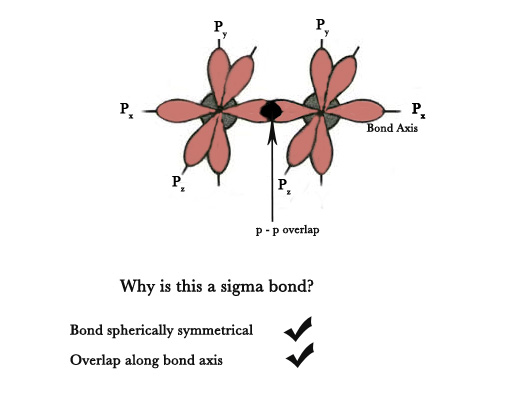

To summarise –
Though VBT was successful in showing us a good picture of how orbitals overlap it was not a foolproof theory. Thus, new theories were proposed in order to explain the bonding in molecules in a better way. We shall discuss the next theories in our successive posts. Till then,
Be a perpetual student of life and keep learning…
Good day!
References and further reading –
1.http://www.chemistry-assignment.com/types-of-overlapping-and-nature-of-covalent-bonds
2.http://www.oneonta.edu/faculty/viningwj/Chem111/Chapters9-10.pdf
Image source –