Elemental carbon in its sp2 hybridized state can form some very interesting structures, which have very wide applications in industry. Fullerenes are one such class of molecules. Fullerenes are allotropes of carbon.
What do you mean by allotropes?
In Greek ,
allos = other
tropos = form.
Allotropes of an element are different forms of the same element with different arrangement of the bonded atoms, in same physical state.
e.g.– Carbon has many allotropes namely – diamond , graphite, graphene , carbon nanotubes and fullerenes. In diamond, all carbon atoms are sp3 hybridized. Thus, in diamond the carbon atoms are in a tetrahedral arrangement. In graphite , the carbons are sp2 hybridized and they occur in layer form. Graphene is a single layered sheet of carbon atoms and carbon nanotubes are one layer tubes of carbon atoms. Fullerenes have carbon atoms in hollow sphere form.

Discovery of fullerenes.
We all have seen structures like these – 
These structures were named ‘geodesic domes‘ by an architect called Buckminister Fuller. Fullerenes are named after this architect and their structure resembles these domes.
In 1984, Sir Harry Kroto and Robert Curl started working on making carbon clusters. They wanted to use Richard Smalley’s laser- supersonic cluster beam equipment. Mr. Smalley was initially not very interested but after a year he decided to work with Kroto and Curl .Harry Kroto packed his bags and moved to Houston from England , to work on carbon clusters. Four days later, after Kroto landed in Houston, Kroto started his work of laser vaporizing graphite with laser beam. After 10 days of rigorous experimentation, they found a peak in mass spectrometer at 60 and 70 carbon atoms! (We will learn mass spectroscopy in detail when we discuss spectroscopy later).
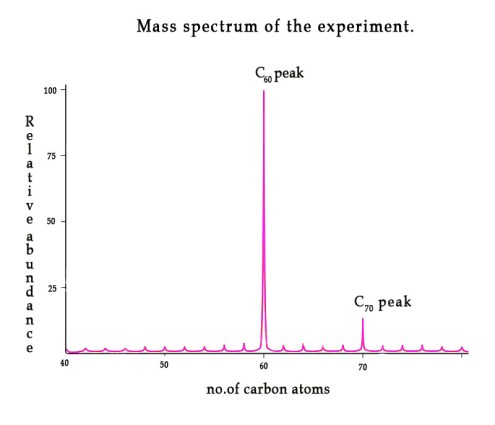
This result was quite unique. This was how the most famous fullerene – C60 was discovered. It was named buckministerfullerene because of its similarity to geodesic domes. It is also referred to as a ‘buckyball‘ by chemists.The diameter of buckyball is around 7Å. In 1996, Nobel prize in chemistry was awarded to these three scientists for their work on carbon clusters.
What are fullerens?
Fullerenes are sphere-like and cage- like molecules of carbon atoms which look like a geodesic dome. Fullerenes consist of fused pentagons and hexagons. No two pentagons lie side by side. They have soccer ball like structure.

The smallest , most stable fullerene obtained by usual preparation methods is the buckyball or C60 – 3D picture of buckyball
C70 has an ellipsoidal cage-like structure. The red atoms indicate five hexagons additional to the C60 molecule.

According to Euler theorem, to close into a spheroid, a fullerene must have exactly 12 five-membered faces, but the number of six-membered faces can vary widely. This is the building principle of fullerenes. The pentagons help in introducing curvature in the molecule.
C60 ⇒ 20 hexagonal faces + 12 pentagons.
C70 ⇒ 25 hexagonal faces + 12 pentagons.
Aromaticity of Fullerenes – Spherical aromaticity.
Each carbon atom in a fullerene is sp2 hybridized and forms σ bonds with three other carbon atoms. The fourth electron is delocalized into the system. The five membered rings are antiaromatic and six membered rings aromatic. Thus, fullerenes have both stabilizing and destabilizing elements in its structure. So, it is very difficult to explain aromaticity of fullerenes.
Aromaticity of fullerenes is not the same as that for benzene or other planar compounds. Fullerenes have a conjugated π system of electrons. They are resistant to high temperatures. They have stable structures. However , some characteristic reactions for planar aromatic compounds are not observed for fullerenes. For example, the typical aromatic substitution reactions are not possible for fullerenes.
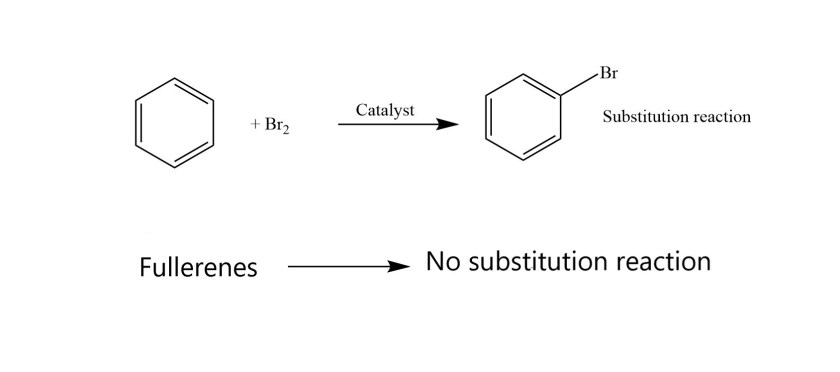
This is because fullerenes are spherical in shape and they don’t have boundaries which are saturated with hydrogen atoms i.e they don’t have hydrogen atoms that can be substituted!
![]()
Fullerenes have only carbon atoms in their structure and no hydrogen atoms.
Fullerenes show addition reactions rather than substitution reactions. Thus, they behave as a ‘polyolefin’ i.e a molecule with many double bonds. However, most of the addition reactions are reversible. This shows the tendency of fullerenes to retain their original stable structure. This can be considered as a major criteria for spherical aromaticity.
In general, a fullerene is aromatic if it had 2n2+2n+1 π-electrons. C60 does not fulfill this rule and so is not aromatic.
Understanding aromaticity of fullerenes is a very complicated subject and beyond the scope of our discussion for now. In the next post, we shall we will talk more about antiaromaticity. Till then, be a perpetual student of life and keep learning…
Good Day !
References and further reading –
1.Fullerenes Chemistry and Reactions by Prof.Andreas Hirsch and Dr.Michael Brettreich
2.https://en.wikipedia.org/wiki/Spherical_aromaticity
Image source –
1.By Pd1000 at English Wikipedia, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=48597530
2.By Science History Institute, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=30097201
3.By Oak Ridge National Laboratory – http://www.ornl.gov/doe/doe_nsrc_workshop/summary.htm, Public Domain, https://commons.wikimedia.org/w/index.php?curid=15196844
4.https://guardian.ng/property/architects-plan-geodesic-dome-skyscrapers-in-lokoja/
5.By Ptj – Polish Wikipedia, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=4431517


