The contention between Albert Einstein and Neils Bohr was carried forward and taken to a new level by their respective students – Erwin Schrödinger and Werner Heisenberg, who were hell-bent to prove that their theory about the atom was correct.
Let us begin talking about these very talented men, who went head-to-head to prove that their line of thought was inerrant.
Erwin Schrödinger –

Erwin Schrödinger was an Austrian physicist, who is credited with developing ‘Schrödinger’s equation – a mathematical equation that can explain the behavior of all systems (macroscopic, sub-atomic, atomic, molecular) in the universe!
He was known to be an attractive, charming, suave, and promiscuous individual. He was a romantic at heart and had a very passionate personality.
In 1925, Schrödinger was getting burned out by his research work, so he decided to go out for Christmas vacation. He went to the Alpine resort in Switzerland with his former girlfriend. He returned two weeks later, with his equation of wave mechanics!! This equation was amazing as it could describe every element in the periodic table!
Erwin Schrödinger took de Broglie’s idea and took it further. He formulated his thesis by considering that the electron was itself a wave of energy. This wave vibrated so fast around the nucleus that it looked hazy- like a cloud of energy.
So, the exact position of an electron could not be determined, although the probability of finding the electron could be calculated.
What does the probability of finding an electron mean?
Probability is a mathematical concept that gives us a method of calculating the chance of a given outcome.
e.g. If we toss a coin, how likely is it that we get heads? Or how probable is the outcome of getting heads? Thus, mathematically we ask a question – what is the probability of getting heads? A coin has only two sides- heads and tails. So, the chances of getting heads after flipping the coin are half i.e 50%. Thus, the probability of getting heads is 50%. Thus, we cannot know what exactly we get after tossing the coin, but we can calculate the probability of an event occurring.
The higher the probability, the higher the chance of that event occurring. The lower the probability, the lower the chances of the event occurring.

Where would you find your mother at 3am every day? At this hour she must be definitely sleeping. So, the probability of finding her in the bedroom is very high. However, what if she got thirsty and went to the kitchen to drink some water? Then she could be in the kitchen as well but the probability of that happening is very low.
Similarly, Schrödinger stated that only the probability, of the location of an electron, can be calculated. The exact location of the electron was not definite. The distribution of these probabilities, around the nucleus, formed areas of space called orbitals. Thus, the probability of finding the electrons in the region of the orbitals is very high (as shown in the figure below).

An orbital is a wave function describing the state of a single electron in an atom. He proposed an equation for wave function, as follows –
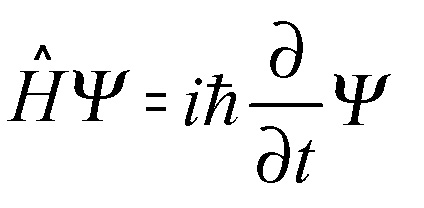
H ⇒ Hamiltonian operator
ψ ⇒ Wave function
i ⇒ Imaginary number
ħ⇒ Reduced Planck’s Constant = (h/2π)
∂/∂t ⇒ Partial differentiation symbol.
It can also be represented as –

E ⇒ Binding energy i.e the energy that binds electrons to the nucleus.
Just as we use Newton’s equations to represent what happens to a ball when kicked, we use Schrodinger’s equation to understand the behavior of sub-atomic particles. (This is because Newtonian mechanics is not applicable at the sub-atomic level). Let us try to understand these terms one by one –
THE OPERATOR CONCEPT IN QUANTUM MECHANICS –
- An Operator is a symbol for a certain mathematical procedure, which transforms one function into another function. So, basically, it means that we are operating on an expression and changing it to another expression.
∴ Operator(function)= Another function. - It is a mathematical procedure or an instruction to carry out certain operations.
e.g.- a square root sign ‘√’, is an operator. If we plug in a value in it, it will simply take the square root of that number and give us a new number e.g. √9 = 3. So the square root sign is an instruction given to a number. Similar instructions can be given to functions as well. An operator has to operate on a MATHEMATICAL FUNCTION. - a symbol that represents a particular mathematical operation being carried out.
- The symbol for an operator is ∧.
- An operator has no meaning unless some quantity is put into it.
e.g. – 1)Square root ‘√ ‘ by itself has no meaning. When a value is put in it only then does it get its significance? √ 16=4.
2)d/dx is an operator.It transforms a function into its first derivative w.r.t ‘x’.
d/dx(xn) = n xn-1 - In Quantum Mechanics all operators are linear.

- In quantum mechanics, physical properties of classical mechanics like energy, time, linear momentum, angular momentum, etc are expressed by operators.
- The Hamiltonian operator is an operator corresponding with the total energy of the system i.e it is associated with the kinetic and potential energies at the sub-atomic level. When the Hamiltonian operator operates on the wave function Ψ(Si), we get Schrodinger’s equation.
THE WAVE FUNCTION (Ψ) –
- It is a mathematical model/function, which represents a wave equation. A wave equation describes the properties of the waves and the behavior of fields. Thus, Ψ is a mathematical description of the particles, which are behaving as waves, at the sub-atomic level.ψ represents a field of some quantity that exists at all points in space.
e.g. Field of temperature on the surface of the earth,ψtemp. The temperature varies in different parts of the world. A wave equation in ψtemp would describe temperatures in different parts of the earth.
- ψ has all the measurable information about the particle. We already know that at the sub-atomic level, particles behave as waves. Thus, Ψ will give us all the wave-like information the particle exhibits. Thus, Ψ is the description of the quantum mechanical state of a particle.
- We know that according to Schrödinger, we just get probabilities at the sub-atomic level. It is a wave equation in terms of the wave function which predicts analytically and precisely the probability of events or outcomes.
- The wave function describes the position and state of the electron and its square gives the probability density of electrons. It describes the quantum state of a particle or set of particles.
Ψ 2⇒Ψ, Ψ* ⇒Probability of finding an electron in unit volume.
Ψ = wave function which has no physical interpretation and could be real or imaginary.
Ψ*=the complex conjugate of Ψ.
Ψ 2 is called the probability density of finding a particle in a particular region.
When Ψ 2 is large → high probability of finding the particle in that region
When Ψ 2 is small → low probability of finding the particle in that region
- Thus, in Quantum Mechanics, the wave function describes the state of a system by the way of probabilities.
- When the operator operates onto the wave function, it extracts all the desired information from it. This information is called the EIGEN VALUE of the observable quantity.OPERATOR(WAVE FUNCTION) = EIGENVALUES.
The Nobel Prize in Physics 1933 was awarded jointly to Erwin Schrödinger and Paul Adrien Maurice Dirac “for the discovery of new productive forms of atomic theory”
Schrödinger’s equation can only be solved for very simple species like-
i) Particle in a box
ii)Harmonic oscillator
iii)Rigid rotator
iv) 1 electron systems.
Beyond these one has to use approximation methods to solve the equation. The methods commonly used are –
1)Perturbation method
2)Variation method.
Understanding the Schrödinger equation is a very complicated process. As students of chemistry, we lack the understanding of the mathematical modeling of particles. Thus, we are only expected to understand some basic ideas of this equation. It is impossible to comprehend this equation/ quantum mechanics in totality without an in-depth knowledge of mathematics. Thus, we just try to discuss the chemical aspects of this esoteric subject. We shall discuss this equation in greater detail when we start our discussions on Quantum Mechanics. For now, understanding these basic parameters would suffice. In the next post, we shall start talking about another genius who was Schrödinger’s rival.
Till then,
Be a perpetual student of life and keep learning…
Good day!
References and further reading –
1)http://timelineoftheatom.blogspot.in/p/erwin-schrodinger-1926.html
2)http://hyperphysics.phy-astr.gsu.edu/hbase/quantum/schr.html
3)http://physics.stackexchange.com/questions/249239/what-is-a-wave-function-in-simple-language
4)https://www.youtube.com/watch?v=gn5vKp09Pfo
5)MIT lecture 6,3.091 by Professor Sadoway.
6)https://www.nobelprize.org/nobel_prizes/physics/laureates/1933/
Image source –
1)By Nobel foundation – http://nobelprize.org/nobel_prizes/physics/laureates/1933/schrodinger-bio.html, Public Domain, https://commons.wikimedia.org/w/index.php?curid=6209244
2)https://planet-earth-2017.com/heat-not-from-the-sun/