
Werner Heisenberg, was a German theoretical physicist,who was studying under Sommerfield.Sommerfield knew about Heisenberg’s interest in Niels Bohr’s thesis and so he introduced him to Bohr’s idea of Quantum Mechanics.Later,Heisenberg started working with Niels Bohr in 1924.He completely supported Bohr’s idea of the atom.
Heisenberg was a very young ,competitive man.He was exceptionally talented.At the age of 22 , he got his PhD under Sommerfield!! Erwin Schrodinger and Werner Heisenberg hated each other’s ideas.Heisenberg took Schrodinger’s ideas as a personal affront.He was now determined to prove that the capricious atom could not be modelled simply as waves(as proposed by Schrodinger) and that just an equation could not reveal its secret.
In 1925, Heisenberg was suffering from hay fever, due to which his face was swollen up beyond recognition.He decided to escape to an isolated island in Norway to spend some time alone .Here he kept thinking about the atom .He decided to give up all mental pictures of an atom and to try to explain the structure of an atom mathematically.To his surprise, he found out that the behaviour of an atom could not even be explained by pure mathematics! In traditional mathematics, if two constants are multiplied , they yield the same answer e.g.- 7×8 =8×7. This however was not true for an atom.He thus developed a new form of mathematics to be able to describe the atom.After this huge discovery, he was excited as well as scared.He returned to University of Göttingen and worked with Max Born to develop a theory ,which today is called ‘Matrix Mechanics‘.This theory was able to describe an atom mathematically.
Although,Albert Einstein absolutely did not like this idea , Niels Bohr and Pauli loved it! A war ensued between Heisenberg and Schrodinger.They both verbally attacked each other, reprehending each other’s theories.
In 1926, Schrodinger was to deliver a lecture on his equation, in Munich.Heisenberg,who was just 24 years old , went all the way to Munich to confront his rival, in front of the scientific community.At the end of Schrodinger’s lecture, Heisenberg attacked Schrodinger’s theory with a monologue.However, the scientific community supported Erwin Schrodinger’s idea and that left Heisenberg deeply hurt.He then returned to Copenhagen to Niels Bohr.Their confidence was low as the entire scientific community was against them but they never gave up on their belief.
In the year 1936, Bohr and Heseinberg used to argue about Quantum Mechanics, night after night ebulliently.They argued so intensely and devotedly that on one occasion Heisenberg was reduced to tears! And then when Heisenberg looked out of the window of his room in despair, an extraordinary thought suddenly struck him! This thought later came to be known as the Heisenberg’s uncertainty Principle!
Heisenberg’s uncertainty Principle –
“It is impossible to determine both the exact position and the exact momentum of an object simultaneously”
(Δpx) (Δx )≥ h/2π.
Δpx ⇒ Uncertainity in the momentum of a particle(Δpx = ΔV.m)
Δx ⇒ Uncertainity in position.
h⇒Plancks constant.
In the above equation, the uncertainty we talk about is NOT the uncertainty related to the measurement.So, this uncertianty is NOT an error which we encounter.It is a uncertainty fundamentally associated with the particle.It means no matter how accurate our measuring instrument is, no matter how accurately we measure the position, we can NEVER ascertain the exact position while finding out the momentum.
Heisenberg realised there were limitations to knowing an atom.There were parts of it that were unknowable! There is a limit of how much we can find out about the sub atomic world.If we find out how fast an electron is moving (momentum) we cannot be sure of its exact position and if we determine its exact position at a given instant, we cannot know how fast its moving.
This can be somewhat explained by an analogy of a small child running around the house.This child is moving in varied speeds in the room.When we try to find the exact position of the child , at that instant we cannot be sure of its speed (as speed is a parameter to be studied over a period of time(Speed =Distance/Time).If we go to determine its speed over some time,in that time its position has already changed as the child is moving.So, both position and momentum cannot be determined simultaneously.
At the sub atomic world , there are more constraints in our ability to observe things.Why ?Let us try to find the answer to this question.
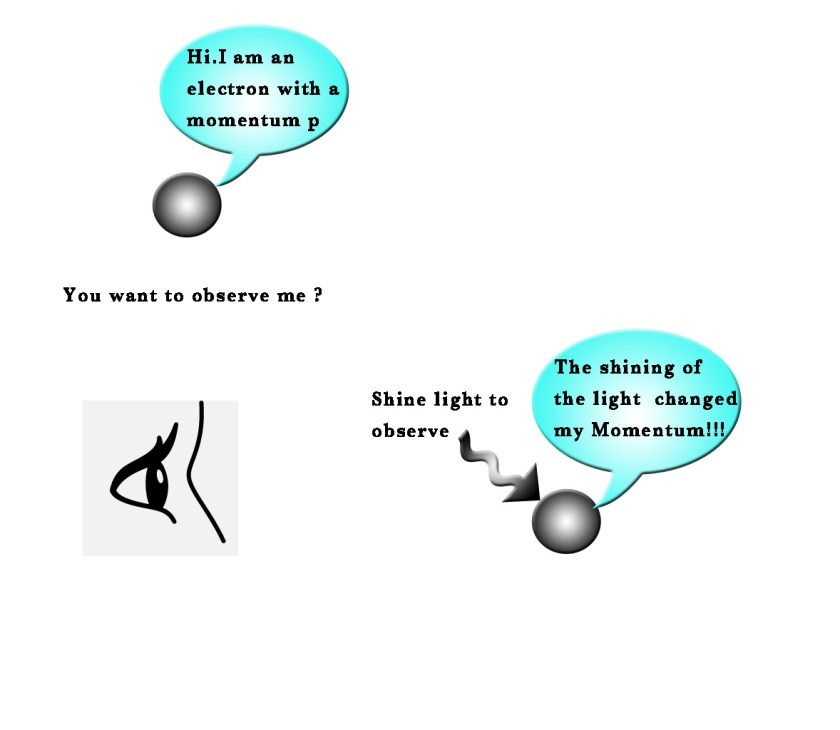
The sub atomic particles, e.g. an electron, moves so fast that it becomes difficult to know its exact position.Also, to see the movement of such particles, we first have to shine light on them. We can see everyday things in light because the photons of light bounce of the surface of things and enter into our eyes.These photons carry specific information about the object/s we see and our brain later processes this information.Now , at the sub atomic level, when we shine light on the system,that we are studying, we are actually transferring some energy to that system (as light is a form of energy ) and thus the photons of light happen to disturb the very system we are trying to study.e.g. – If we shine light on an electron, the photons of that light will transfer energy to the electron and thus it will start moving with a greater speed .The photon will invariably impart some momentum to the electron under study.Thus, its momentum will change and we thus cannot be sure of what its initial momentum (before we shine light on it) was.
Niels Bohr realised that the act of observing, changes the outcome of the experiment!He proposed that before one observes an experiment, its outcome is uncertain.So, for the double slit experiment, we observe the electron (after passing through the double slit) at some point on the screen.Just before the moment when we observe the result, the electron (which is behaving as wave ) could be practically anywhere in the space which defines the wave.Only at the instant where we observe it on the screen do we get a fixed position of that electron at that moment.So our act of observing at that moment gives it a definitive state.So, the very act of observing, forces the particle(electron) to make that choice.
With this theory,everything fell neatly into place for Niel’s Bohr and Werner Heisenberg.They concluded that the atom can be completely described only by pure mathematics!
In October 1927, at the fifth Solvay International Conference , in Brussels, Niels Bohr decided to put forth his discovery in front of all the notable physicists.This conference was perhaps the most exciting one ,for all leading atomic physicists at that time attended it to talk about the Quantum Theory.Everyday Albert Einstein questioned Niels Bohr’s ideas and Bohr would answer them and brush those remarks aside.By the end of the conference, Bohr became victorious and Quantum Mechanics was accepted by the entire scientific community to be true! This was the only time in history when so many great physicists were seen together.

This was the moment in Science when the deterministic models(models considering individual entities) were changed into probabilistic models(particle ensembles)!The Copenhagen interpretation(Niel’s Bohr) is still accepted today!
Inspite of the rivalry between the two scientists, Erwin Schrodinger and Werner Heisenberg,their theories just complimented each other and are still considered as major breakthroughs in Quantum Mechanics!
The entire world as we know today ,including all the electronics we use, is a result of this Quantum Mechanics!Our world would just collapse without Quantum Mechanics! I intend to discuss Quantum Mechanics in much greater detail later.In our next post, we shall talk about some more, recently discovered, sub atomic particles.Till then,
Be a perpetual student of life and keep learning!
Good day !
Image sources –
1)https://upload.wikimedia.org/wikipedia/commons/thumb/f/f8/Bundesarchiv_Bild183-R57262,_Werner_Heisenberg.jpg/220px-Bundesarchiv_Bild183-R57262,_Werner_Heisenberg.jpg
2)http://photos.aip.org/favorites.jsp
3)http://newvaluestreams.com/wordpress/?p=3418
4)By Benjamin Couprie – http://w3.pppl.gov/, Public Domain, https://commons.wikimedia.org/w/index.php?curid=21332727
References and further reading –
1)http://www.nobelprize.org/nobel_prizes/physics/laureates/1932/heisenberg-bio.html
2)https://en.wikipedia.org/wiki/Uncertainty_principle
3)Lecture 6, MIT open courseware,3.091 by Professor Donald Sadoway.
4)Atom: The Clash of the Titans – BBC Documentary (PART1)