Let us begin our discussion on one of the most profound branches of chemistry. This branch almost exclusively deals with only one element and its compounds- CARBON! Organic chemistry is all about CARBON and CARBON COMPOUNDS.
In organic chemistry, we study the structure, properties, and reactions of organic materials and compounds i.e species containing carbon atoms in their structure.
e.g.- Shown below are the structures of some organic compounds- n-propane, benzene, furan, and methane.

Note – The cyclic structures of benzene and furan have a carbon atom at each corner of the structure (except where O is present) as shown below-
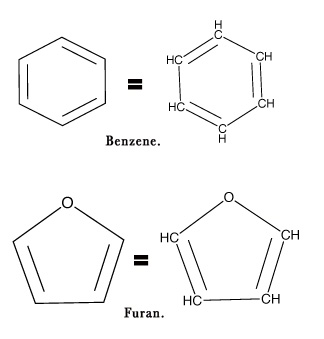
We shall begin studying the very basics of organic chemistry and later start discussing different chapters in detail. So, lets us begin our journey into this amazing world of organic molecules!
Catenation.
What is so special about the element carbon? Why do we have so many carbon compounds? Why does the element carbon get an entire branch of chemistry dedicated to its study? The answer to these questions is a property called ‘catenation‘.
The property of self-combination of atoms of the same element to form chains or rings is called catenation.
Catenation is a property of binding of an element to itself through covalent bonds to form straight/branched chains or ring molecules. Thus, the self-combination of atoms of the same element to form chains or rings is called catenation.
The carbon atoms can self-link to each other to form carbon chains or carbon rings. Why does the carbon atom exhibit this unique phenomenon?
It is because each carbon atom has 4 valence electrons, which can be shared with another carbon atom to form a carbon-carbon single bond.
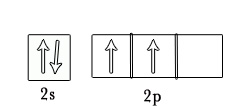
Carbon 1s2 2s2 2p2
Carbon atoms can bond with each other covalently in various ways to form a very wide range of compounds. This property is what makes carbon chemistry so unique and vast.

As seen in the above figure, 8 carbon atoms can be arranged in various ways to form different compounds. Thus, the possibilities for forming different carbon compounds are limitless. This makes the scope of organic chemistry very vast.
General characteristics of organic compounds –
- All organic compounds predominantly contain carbon and hydrogen. Other elements found in organic compounds are oxygen, chlorine, bromine, nitrogen, sulfur, etc.
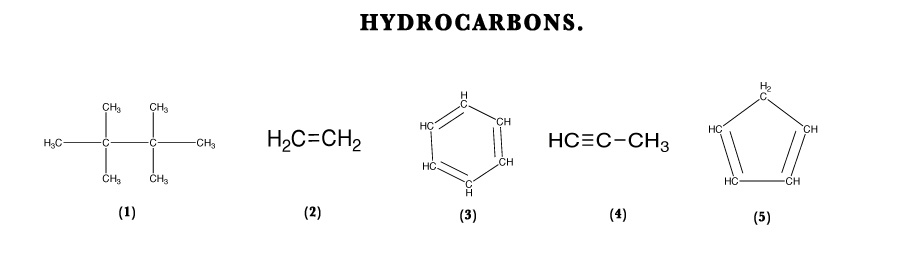
Compounds that contain only carbon and hydrogen are called ‘Hydrocarbons‘.
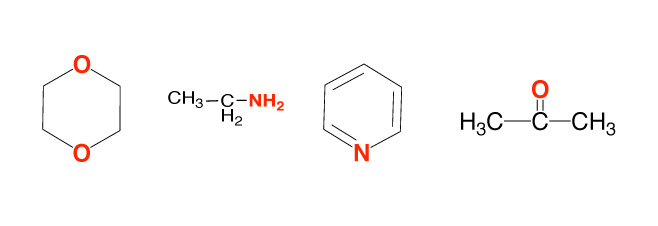
- The chemical behavior of organic compounds depends on the presence of certain atoms or groups of atoms called the functional group. Compounds with similar functional groups have similar properties.e.g.-Amino group (-NH2 ), a carboxyl group (C=O), alcohol group (-OH).

3. Organic compounds are generally covalent and thus are insoluble in water. However, they are soluble in organic solvents like ether, benzene, ethanol, etc.
4. Many of them have low melting and boiling points. Many are volatile.e.g.-Acetone.
5. Organic compounds are flammable.
6. They are less reactive than ionic compounds
Examples of organic compounds-








We encounter innumerable organic compounds in our day-to-day lives. We as humans are organic beings!! Our body is made up of many organic molecules like proteins, nucleic acids (DNA & RNA), lipids, hemoglobin, hormones, etc. The food we eat contains organic molecules – carbohydrates, lipids, proteins, vitamins, etc
Fuels, cosmetics, dyes, drugs – organic compounds are just everywhere!
Organic chemistry is at the heart of so many things! Let us start delving deeper into this amazing world of carbon compounds and find out all these molecules in our successive posts. Till then,
Be a perpetual student of life and keep learning…
Good Day!
References and further reading –
1.https://www.thoughtco.com/definition-of-catenation-and-examples-604886
Image source –
1.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwiH0e3Vh-fTAhXFLo8KHVvUBAoQjRwIBw&url=http%3A%2F%2Fwww.kidneycoach.com%2F253%2Fhow-to-increase-hemoglobin%2F&psig=AFQjCNG7IwX3VVZm6kF2Ated_JL2L1j_Fg&ust=1494565259652056
2.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwi-sJ6oiOfTAhUaSI8KHdaqD6AQjRwIBw&url=http%3A%2F%2Fwallpapersfan.com%2F3d_dna_structure_molecule_pattern_abstraction_gene_wallpaper-74&psig=AFQjCNH4H2B4GTNS8AsrVn9A_qOs4IlAgg&ust=1494565389459441
3.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwjli8bKi-fTAhUKPY8KHWIpBzcQjRwIBw&url=https%3A%2F%2Fwww.glutenfreegigi.com%2Fgluten-in-otc-and-prescription-medicines%2F&psig=AFQjCNFuLTVRhfbCiuFz00iPg8xppSCMHw&ust=14945663149362314.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwiesaTqi-fTAhUHSI8KHUuxDncQjRwIBw&url=http%3A%2F%2Fwww.andhrawishesh.com%2F378-telugu-headlines-top-stories%2F54138-petrol-and-diesel-rates-hiked.html&psig=AFQjCNHVpajX0wlZwhJ4bBRN4KE_iQRXBQ&ust=1494566380645624
5.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwi5zt2cjOfTAhWKr48KHfFWCXUQjRwIBw&url=http%3A%2F%2Fwww.swatchandlearn.com%2Fbeauty-secrets-acetone-nail-polish-remover-review-pictures%2F&psig=AFQjCNGNDsWspcrRByH–38H_V4ohW_VCQ&ust=1494566495172147
6.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwjevMq2jOfTAhVFQY8KHcUECM0QjRwIBw&url=https%3A%2F%2Fwww.applyiec.in%2Fregistration-imports-cosmetics-india%2F&psig=AFQjCNGeuOgfYbvaKaHfpWgZHes93U5IVQ&ust=1494566524391833
7.https://www.google.co.in/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0ahUKEwiOkvvhjOfTAhXBPI8KHRqDApwQjRwIBw&url=http%3A%2F%2Fblessedbeyondcrazy.com%2Fpros-and-cons-of-various-cookware%2F&psig=AFQjCNEbJUo6RvO31NFCGMsmN4M6iYT7UA&ust=1494566619080764