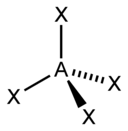
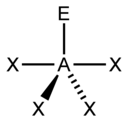
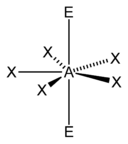
In the VSEPR model, the valence shell is considered a sphere. The electron pairs are considered to be on this spherical surface, as far away as possible from one another. The maximum distance between the electron pairs ensures that there is minimum repulsion between them.
In our last post, we discussed the geometries of molecules without lone pair of electrons. However, due to the presence of non-bonding / lone pair of electrons, molecules depart from ideal electronic geometry/bond angles. Let us discuss how these non-bonding pairs affect molecular geometries.
The effect of non-bonding/lone pair of electrons.
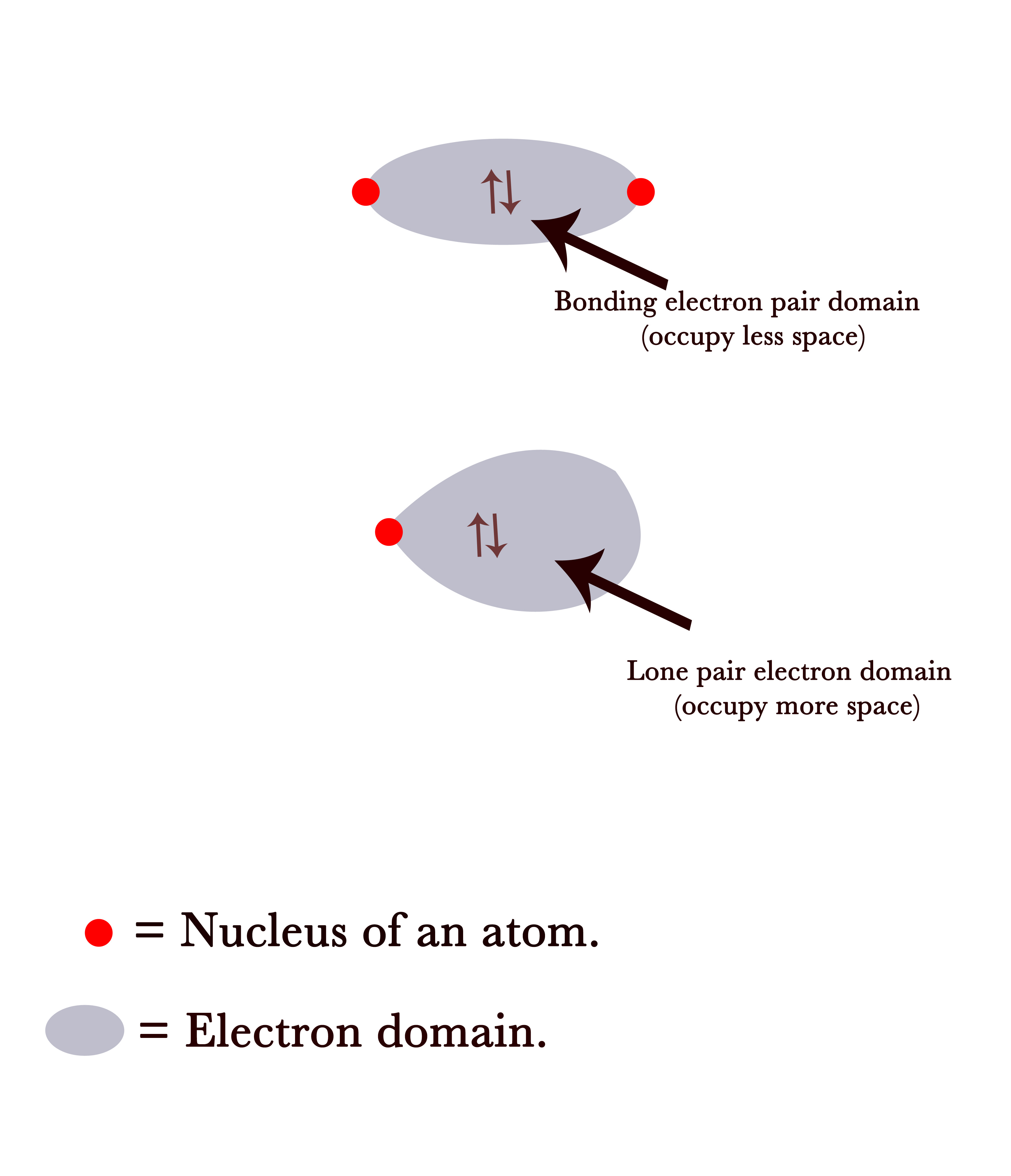
The VSEPR model predicts that the presence of lone pair of electrons in a molecule results in a deviation from ideal geometries/ bond angles. Such deviations can be explained by the electron domain concept. Electron domains are the regions in space occupied by an electron pair.
The reasons for deviation from ideal geometries are –
- Non-bonding/lone pairs occupy more space than bonding pairs –
The bonding pair of electrons are a part of a bond between two atoms. Thus, they are diffused through the orbitals of both A and B and between the nuclei of both atoms. Naturally, they are away from the nucleus of one single atom and are equally attracted by both nuclei. This leads to a decrease in the electron domain size.
A lone pair of electrons is on a single atom and is only attracted by a single nucleus. So having a positive nucleus only at one end keeps the electron domain “fatter” as compared to that of a bonding pair domain.
2. The intensity of repulsion between different electron pairs is different.
It is as follows –
Lone pair – Lone pair > Lone pair – Bonding pair > Bonding pair – Bonding pair electrons, i.e
LP – LP > LP- BP > BP – BP.
As the lone pairs occupy more space, the electron domains of two lone pairs on a single atom come close to each other.Thus, the repulsion between them is more. This is true for LP-BP interactions too.The BP electron domains are the farthest apart from each other and thus there is minimum repulsion between them.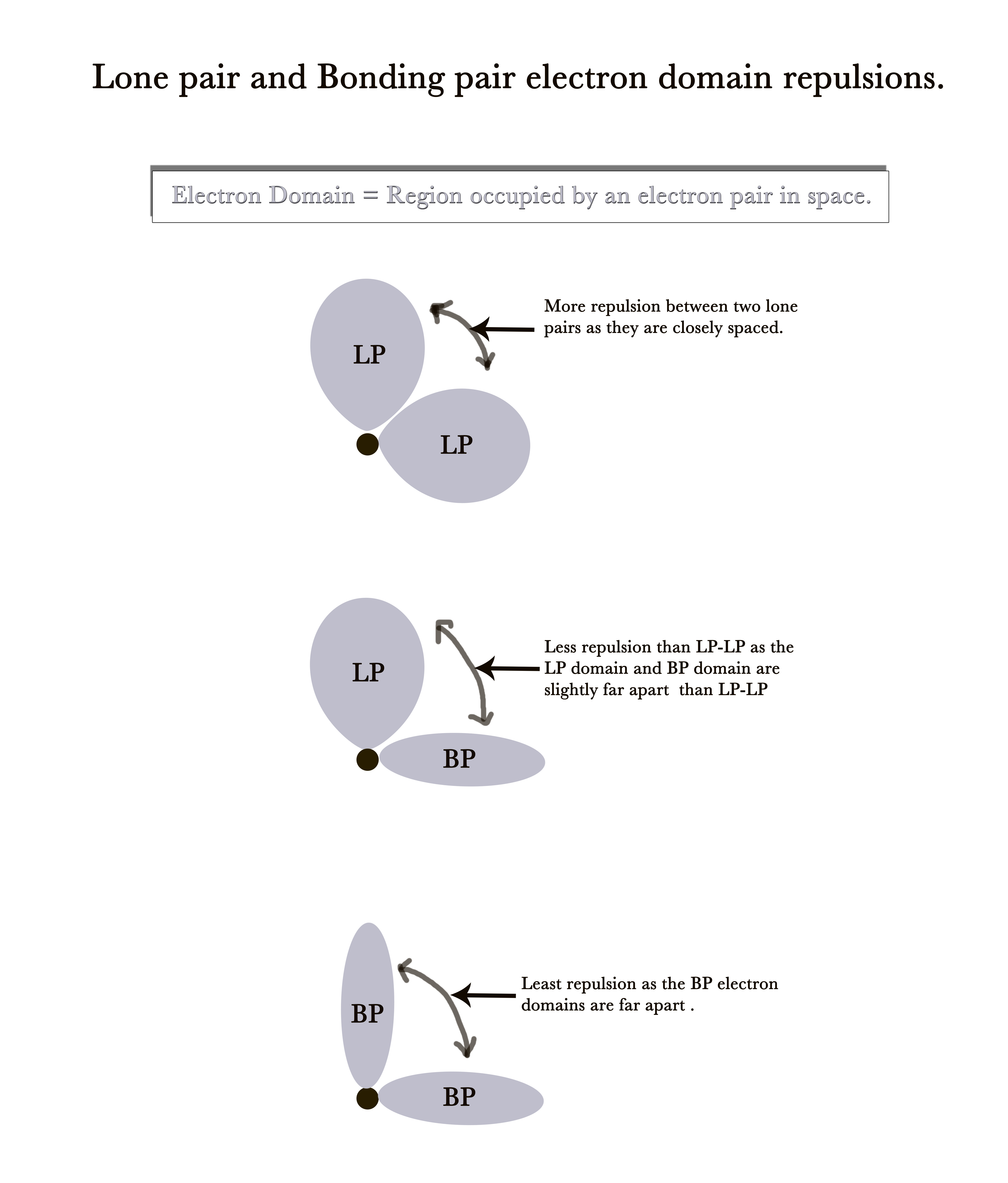
3. Repulsions occurring at angles more than 90 °are not significant.
4. The bonding pairs of the electronegative substituents occupy less space than the electropositive ones.
This is because these electrons are strongly attracted by the electronegative atom. Thus, the repulsion between two BP’s becomes less and so the bond angle becomes less.
Let us understand this with an example.
|
Molecule |
Bond angle(H-N-H) |
|
NH3 |
107.2 |
|
NF3 |
102.3 |
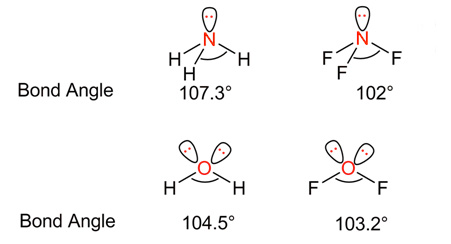
In the above example, fluorine is more electronegative than hydrogen.
So, BP of electrons between N – F has attracted more towards the F atom → repulsion between N-F bonding electron pairs decreases → the N-F bonds come closer to each other as a result of the decrease in repulsion between them → Bond angle decreases.
Think of these electron pairs as individuals! When the hatred(repulsion) between two people decreases, they automatically come closer to each other, and thus the bond angle decreases! Atoms, human beings… it’s all the same, isn’t it?
5. Electron pairs in a filled shell repel stronger than electron pairs in the incomplete/vacant shell.
When there is an vacant shell (of similar energy) in an atom, the electrons from the filled shell can undergo diffusion to these orbitals. The electron pairs in a vacant shell repel less. Hence, the LP – BP repulsion diminishes dramatically. Thus, the bond angles around the central metal decrease.
Less repulsion between LP -BP → the bonds come closer → decrease in bond angle.
Look at the examples below-
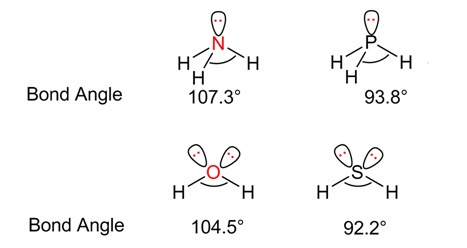
Example 1 gives us the difference between the bond angles in ammonia(NH3) and phosphine(PH3).
The central element in ammonia is nitrogen. N (7) 1s2 2s2 2p3.
The nitrogen atom does not have any d-orbitals and so the 2p electrons have no chance to go to any other orbital.
However, the phosphorous atom has vacant 3d orbitals. P(15) – 1s2 2s2 2p6 3s2 3p3 3d0
The energies of 3p and 3d orbitals have the same energy. Thus, the 3p electrons can easily diffuse to the 3d orbitals. This decreases the repulsive forces and leads to a decrease in the bond angle in phosphine.
Example 2 –
The central atom is oxygen. O(8) 1s2 2s2 2p4
Oxygen does not have any vacant orbitals of the same energy.
Sulfur however has them. S (16) 1s2 2s2 2p6 3s2 3p4 3d0
6. Triple bonds repel other bonding electrons more strongly than double bonds and double bonds repel other bonding electrons more strongly than single bonds.
Based on the above facts let us now study some examples, where lone pair affects the geometry of the molecules –
Deviation from tetrahedral geometry /109.5 °bond angle – We studied in the earlier post, that four electron domains exhibit a tetrahedral geometry with a bond angle of 109.5°. So, methane has a bond angle of 109.5°. However, when lone pairs are present in a molecule, the measure of this ideal bond angle (109.5 °) changes and so does the geometry of the molecules.
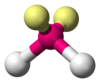
e.g.- Ammonia has a pyramidal shape(bond angle = 107.3 °) with one lone pair of electron domains and water molecule has a bent shape( bond angle = 104.5 ° ) as it has 2 lone pairs of electron domains. This change in geometry is due to the presence of one and two lone pairs of electrons on ammonia and water respectively. The LP occupies the vertices of the tetrahedron.
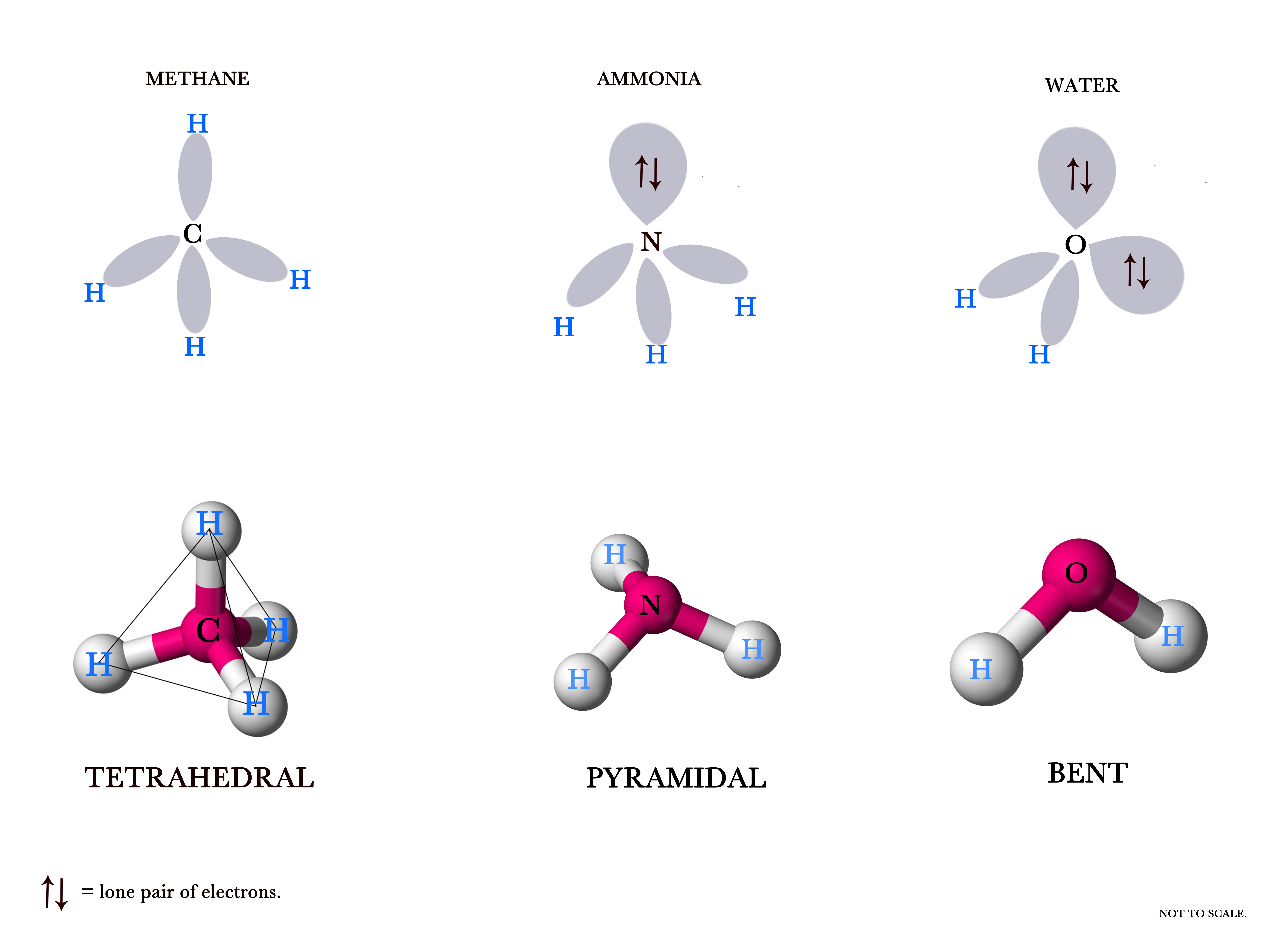
As the lone pair is ‘fatter’, it occupies more space, and thus it pushes the bonding pair domains closer to one another. Thus, the bond angle and geometry change.
(Imagine three slim people comfortably sitting in a car’s rear seat. What happens when one fat person replaces a slim one? The two thin people are pushed closer to make room for this fatter person. A similar thing happens to the electron domains too).
The following table shows the deviations from the ideal geometries –
The above chart shows how the geometry changes with the introduction of one and two lone pairs of electrons on the central atom.
Steric numbers of 7 or greater are less common.
In our next post, we continue the discussion on VSEPR and study some examples. Till then,
Be a perpetual student of life and keep learning…
Good day!
References and Further reading –
1.https://en.wikipedia.org/wiki/VSEPR_theory
2.http://nptel.ac.in/courses/104103069/14
Image source –
1.https://en.wikipedia.org/wiki/Bent_molecular_geometry
3.https://en.wikipedia.org/wiki/VSEPR_theory
4.By Benjah-bmm27 – Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=2035653