Lone pairs – AXIAL OR EQUATORIAL POSITION?
In the structures mentioned in the previous post, we have two positions, namely – axial and equatorial.
Equatorial positions lie on the equatorial plane – the plane with the maximum number of atoms, which lies in the equatorial position (like the equator, in the middle).
Axial positions are perpendicular to the equatorial plane – one above one below the plane.
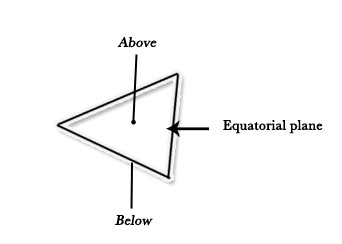
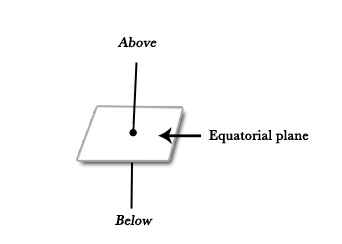
The following figure shows a trigonal bipyramidal structure, where 4 atoms are in the equatorial plane (3 equatorial positions and one central atom) and 2 atoms in axial positions-

If there are two lone pairs of electrons, the question is, which positions will the lone pairs occupy? There are 3 possibilities as shown below –
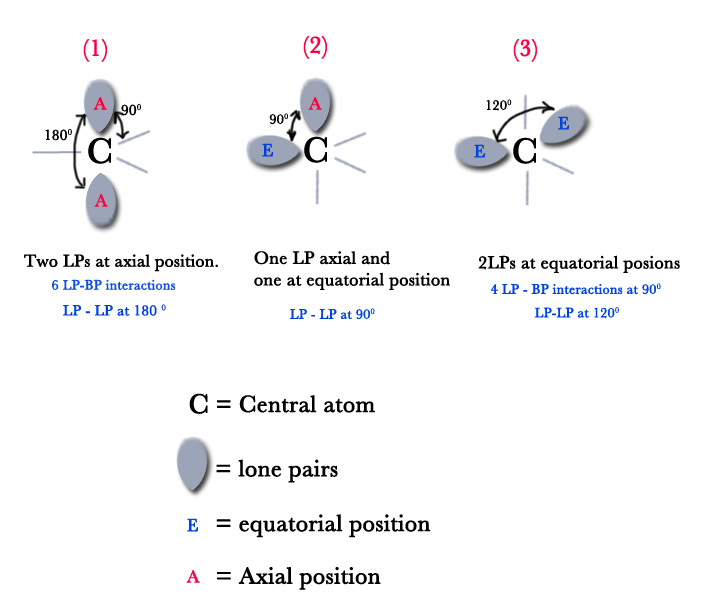
Possibility #(1) – Both the lone pairs(LP) occupy axial positions. In that case, the bonding pairs(BPs) will occupy equatorial positions.
As seen in the figure above, the angle between two LPs is 180º. Thus, they are far apart from each other and the LP- LP repulsion is insignificant (repulsions between substituents that are 90º apart are insignificant). However, in this position, there are 6 LP – BP repulsions. All three BP electrons experience repulsion from both lone pairs as they are only 90º (perpendicular) apart from the bonding pair of electrons. Thus, this possibility is not favored.
Possibility #(2) – One lone pair occupies an axial position and the other one occupies an equatorial position.
In this case, two lone pairs are 90º apart and so the high-intensity LP-LP repulsion is maximum. As molecules tend to avoid maximum repulsions, this case is not favored too.
Possibility #(3) – Two LPs occupy the equatorial positions.
As they are in the equatorial position, they are 120º apart (they occupy corners of an equilateral triangle). Also, there are only 4 LP-BP repulsions. So, this case is favored and thus, LONE PAIRS PREFERABLY OCCUPY EQUATORIAL POSITIONS.
EXAMPLES
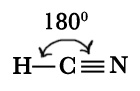
A] HCN.
| Structure: | 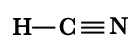 |
| Central atom: |
Carbon, C (6) [He] 2s2 2p2 |
| Valence electrons on the central atom: |
4 |
| H contributes 1 electron, N contributes 3 electrons | 3+1 = 4. However, we consider a triple bond as a single bond so we subtract 4 electrons(2 bonds have 4 electron pairs). ∴8-4 = 4 |
|
TOTAL: |
4 |
| Divide by 2 to give electron pairs |
4/2 = 2 |
| VSEPR geometry(AX2E0) |
Linear |
| Bond angle ( found out experimentally) |
180º |
B] PO43-
| Structure: | 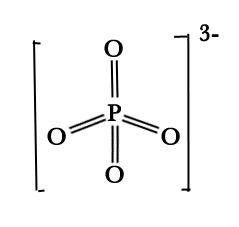 |
| Central atom: | Phosphorous, P(15) 1s2 2s2 2p6 3s2 3p3 |
| Valence electrons on the central atom: |
5 |
| 4 O contributes 2 electrons each, 3 electrons of (-3) charge | 5+8+3 = 16. However, we consider a double bond as a single bond so we subtract 8 electrons(4 double bonds have 8 electron pairs). ∴16- 8 = 8 |
|
TOTAL: |
8 |
| Divide by 2 to give electron pairs |
8/2 = 4 |
| VSEPR geometry(AX4E0) |
Tetrahedral |
| Bond angle ( found out experimentally) |
109.5º |
C] XeOF4
| Structure | 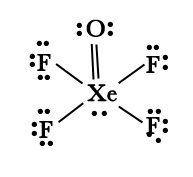 |
| Central atom: | Xenon , Xe (54) [Kr] 4d10 5s2 5p6 |
| Valence electrons on the central atom: |
8 |
| 4F contributes 4 electrons, O contributes 2 electrons, and 2 lone pair electrons on Xe atom | 8+4+2-2 = 12. ( But a double bond is considered a single bond, so subtract 2 electrons. Only LP on the central atom is considered) |
|
TOTAL: |
12 |
| Divide by 2 to give electron pairs |
12/2 = 6 |
| VSEPR geometry(AX2E0) |
Square pyramidal ( not octahedral due to the presence of LP on Xe) |
| Bond angle ( found out experimentally) |
≈ 90º |
Although the VSEPR model is great at predicting geometries for most molecules, it fails for many other molecules, like the transition metal compounds. So, in our next post, we start discussing another very important theory, which is developed on a very different approach. Till then,
Be a perpetual student of life and keep learning…
Good Day!
References and Further Reading –