In the last post we studied some examples of homocyclic aromatic compounds. What happens if the ring has an atom other than carbon in it? Can these compounds be aromatic too? Let us find that out in this post.
Hetero (heteros in Greek) = other (than carbon atom)
Cyclic = ring structure.
Cyclic compounds which have an heteroatom in their ring structure are referred to as heterocyclic compounds
e.g.– 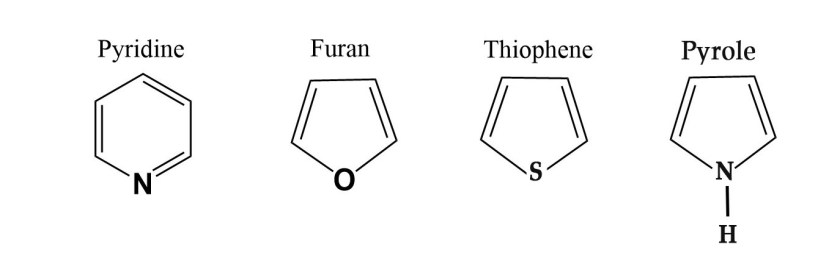
Every ring above has a hetero atom i.e an atom other than carbon atom. N,O,S all are heteroatoms.

In an aromatic system-
i)Nitrogen with no formal charge always has 1 lone pair of electrons.
ii)Oxygen with no formal charge has 2 lone pairs of electrons.
iii)Sulphur with no formal charge has 2 lone pairs of electrons.
iv)Boron with no formal charge has no lone pair of electrons.
So, the above structures can be represented as –
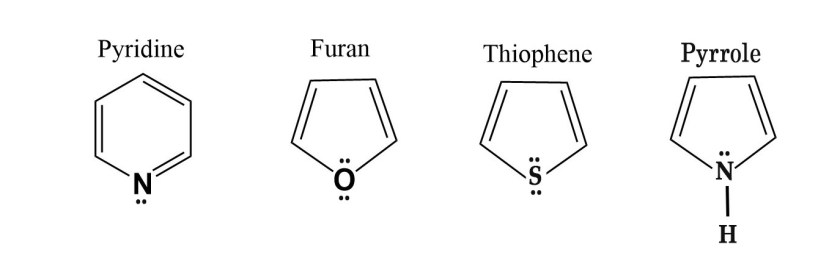
Let us see why the above structures are aromatic.
Pyridine
If one C- atom of benzene with it’s hydrogen is replaced by a N-atom, it gives pyridine molecule. The structure of pyridine (C5H5N) is –

The structure of benzene is similar to the structure of benzene. The differences between the two structures are –
- The C-H bond in benzene is replaced by N in pyridine.
- The C-N bonds are smaller than the C-H bonds,owing to higher electronegativity of N atom. Thus, the geometry of pyridine is NOT a regular hexagon as in benzene.
- The lone pair on nitrogen is NOT involved in forming the aromatic sextet and so is available. Thus, pyridine is basic, unlike benzene.
Nitrogen in pyridine is sp2 hybridized. 
Nitrogen has a lone pair of electrons in one of its sp2 hybridized orbital. The unhybridized p-orbital is a part of the 6 π electron system of pyridine. (Only one sp2 hybridized orbital containing the lone pair is shown below. The other two hybridized orbitals form sigma bonds with adjacent carbon atoms).

The resonance structures of pyridine can be shown as follows –
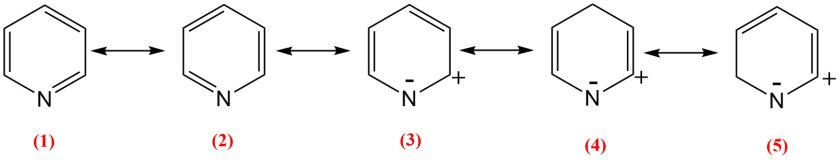
As seen above , the first two structures (1) and (2) are neutral resembling benzene resonating structures. However, structures (3),(4) and (5) have negative charge on N atom. Thus, pyridine is slightly polarised with slight negative charge on N atom and fractional positive charges on α– and γ– positions.

The unhybridized atomic orbital on nitrogen combines with the atomic p-orbitals of five carbon atoms to form six π molecular orbitals as shown below –
 As seen in the above figure, the degeneracy of is Ψ2 , Ψ3 and Ψ4 , Ψ5 lost (benzene had degenerate levels), owing to the distorting effect of the hetero atom, N. The six π electrons are accommodated in three bonding orbitals – Ψ1 ,Ψ2 , Ψ3. Thus, pyridine is aromatic. As it has a lone pair of electron, which is free, it is a base (Base is a species which can donate electrons).Reactions with nucleophiles take place at 2- , 4- and 6- positions. We will study more about the reactions of pyridine when we study heterocyclic compounds.
As seen in the above figure, the degeneracy of is Ψ2 , Ψ3 and Ψ4 , Ψ5 lost (benzene had degenerate levels), owing to the distorting effect of the hetero atom, N. The six π electrons are accommodated in three bonding orbitals – Ψ1 ,Ψ2 , Ψ3. Thus, pyridine is aromatic. As it has a lone pair of electron, which is free, it is a base (Base is a species which can donate electrons).Reactions with nucleophiles take place at 2- , 4- and 6- positions. We will study more about the reactions of pyridine when we study heterocyclic compounds.
In the next post we will study some more examples of aromatic heterocyclic compounds. Till then ,
Be a perpetual student of life and keep learning..
Good day !