For a chemical reaction to occur the reacting molecules must be activated. Usually, this activation is carried out thermally i.e. by increasing the reaction temperature. Increasing temperature results in an increase in the energy of the molecules present in the system. Molecules that have acquired sufficient energy, react and lead to the formation of products. These types of reactions are called thermal reactions.
However, there is another way of introducing energy in a system – by absorption of radiation(light).
Photochemistry is the study of reactions caused by absorption of IR, visible or UV light or which are accompanied by emission of chemical energy as radiation.
The study of photochemistry deals with the interaction between molecules and photons of IR, visible or UV light and the subsequent chemical changes. Thus, photochemistry is chemistry as a consequence of light.
Examples of photochemical reactions.
- Origin of life – Photochemistry is very fundamental to our existence. The sun is one of the main reasons why life exists on Earth. The origin of life must be a photochemical process, as only solar energy was available for simple molecules like methane(CH4) and carbon dioxide (CO2) to react and form complex molecules like proteins and nucleic acids. These molecules are the basis of life.
- Photosynthesis – The primary source of energy for all animals is through food. Plants synthesize various complex organic molecules like glucose, using solar energy. This process is called as photosynthesis. This is a photochemical process too.
- Vision – We see everything in presence of light.The chemistry of vision is a photochemical process too.
- Formation of ozone – The ozone layer in the earth’s atmosphere protects us from the harmful UV rays coming from the sun. The formation of ozone from oxygen in the atmosphere is a photochemical reaction too.
- Bioluminescence – Many organisms like glow worms, fireflies etc emit light from their bodies. This is called bioluminescence.
- Photography – The basis of photography are photochemical reactions too !
- Phototropism – Orientation of plants or animals either towards (positive phototropism) or against(negative phototropism) light.
- Treatment of jaundice in newborn babies – Many newborn babies develop infant jaundice after birth. It is treated by phototherapy, where bilirubin in the baby’s body is broken down using blue light.
- Biosynthesis of vitamin D -Vitamin D is synthesized in our body in the presence of light from the sun.
- Photochromism – The change in colour of glasses when exposed to light of different intensity.
- Photopolymerization – Small molecules (monomers) are converted to polymers using light. These are important photochemical reactions used to make a variety of different materials.
In today’s world, where the energy crisis is becoming a major problem, harnessing solar energy could be an effective alternative to solve this problem. If suitable photochemical reactions are discovered and devices for proper utilization of this abundant source of energy are perfected, half the world’s energy problem can be solved. So let us begin our study on photochemistry to understand this topic better.
Difference between thermal and photochemical reactions
Energy is needed for any chemical reaction to occur. This energy can be either in the form of heat(Thermal reactions) or in the form of light energy (photochemical reactions).
THERMAL REACTIONS – In thermal reactions, the heat provided gets evenly distributed among all molecules. The heat leads to increase in the number of collisions between the reactant molecules and walls of the container. Thus, the vibrations in molecules increase. Molecules have rotational and vibrational and electronic energy levels. This absorbed heat energy leads to excitation in vibrational and rotational levels only. The electronic levels are far apart in energy(as seen in the figure below) and so thermal energy generally DOES NOT bring about electronic excitation. The heat leads to increase in the number of collisions between the reactant molecules and walls of the container.
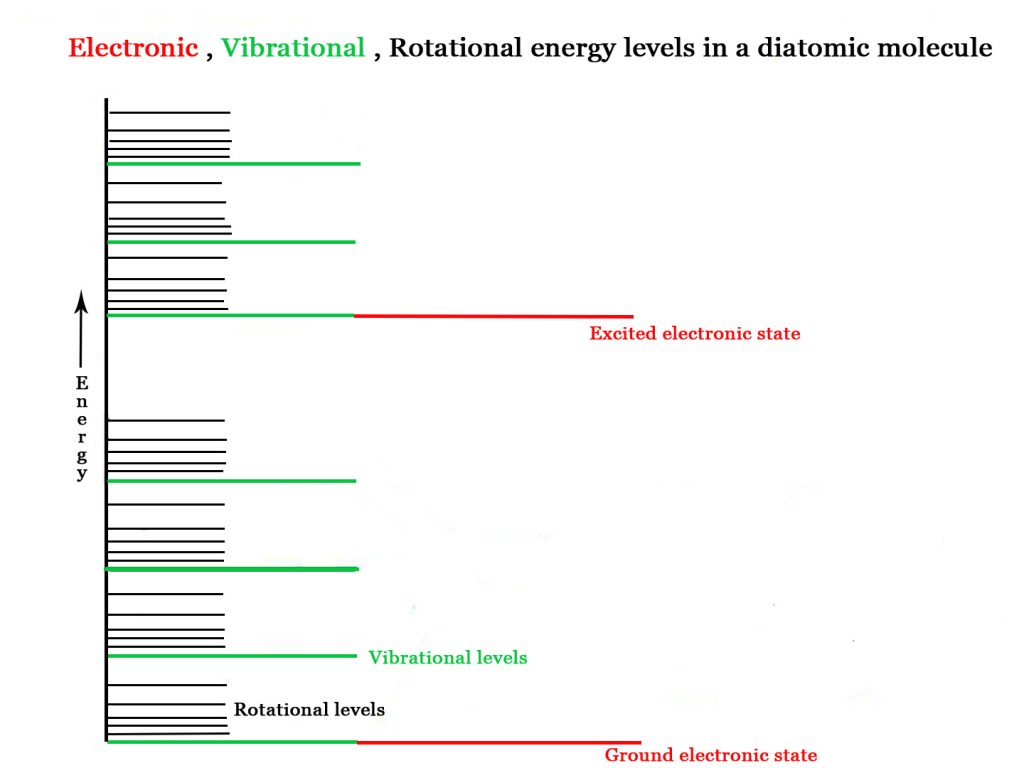
Many reactions which cannot be carried out thermally, can be successfully carried out using electromagnetic radiation. What is the fundamental difference between thermal and photochemical reactions? In photochemical reactions, we can selectively excite specific reactants to get particular products. Thermal reactions lack this kind of selectivity.

As seen in the earlier post, different wavelengths of light have different energy. Wavelength and energy are inversely proportional to each other i.e if the wavelength(λ) of light is shorter, it has higher energy and vice versa.

When reactants react there is a change in their electronic structure. We know that electrons in different orbitals have different energies. So, in a photochemical process, if the light of a specific wavelength is shone on the reactants, ONLY ELECTRONS THAT HAVE ENERGY EQUIVALENT TO THAT OF THE INCIDENT LIGHT WILL ABSORB AND GET EXCITED TO GIVE SUBSEQUENT PRODUCTS. Thus, we can selectively choose our radiation to produce the desired reaction.
Photochemistry and radiation chemistry.
These two terms – photochemistry and radiation chemistry are different from each other.
Photochemistry is a term used to study reactions from IR, visible and UV radiation.
Radiation chemistry deals not only with photochemical processes but also with many more reactions brought about by other high-energy radiations like α, β, γ, cyclotrons, X-rays, or corpuscular beams like neutrons, protons or α-particles(obtained from nuclear reactions or electric accelerators). etc.
Thus, photochemistry is small branch of radiation chemistry which deals with low energy radiation in UV, visible and IR range. In our discussions on photochemistry we will be exclusively talking about reactions that use UV, visible or IR light to convert reactant to products.
In the next post we will try to understand the fundamental difference between these two kinds of reactions. Till then,
Be a perpetual student of life and keep learning….
Good day !