In this post, we shall start learning about the basic laws of photochemistry. These laws form the basis of all the photochemistry we will study later.
The first law of photochemistry
Light incident upon a species can be reflected, refracted, transmitted, scattered, or absorbed. Prior to 1817, photochemical changes like the photofading of colored materials or photosynthesis were studied only qualitatively. It was Christian von Grotthus (1785-1822) and John Draper (1811-1882), who put forth the quantitative aspect of it. They gave us the first law of photochemistry – the ‘Grotthus-Draper law‘.
Only that light which is absorbed by a system can cause chemical change.
Grotthus -Draper law.
As studied earlier, radiation is absorbed only if its energy matches with the energy of the electrons in the reactant species. Thus, each chemical substance absorbs only certain wavelengths of radiation, the set of which is unique to that substance. Thus, the incident light is NOT effective in bringing about a chemical change, if it is NOT absorbed by the reactant species.
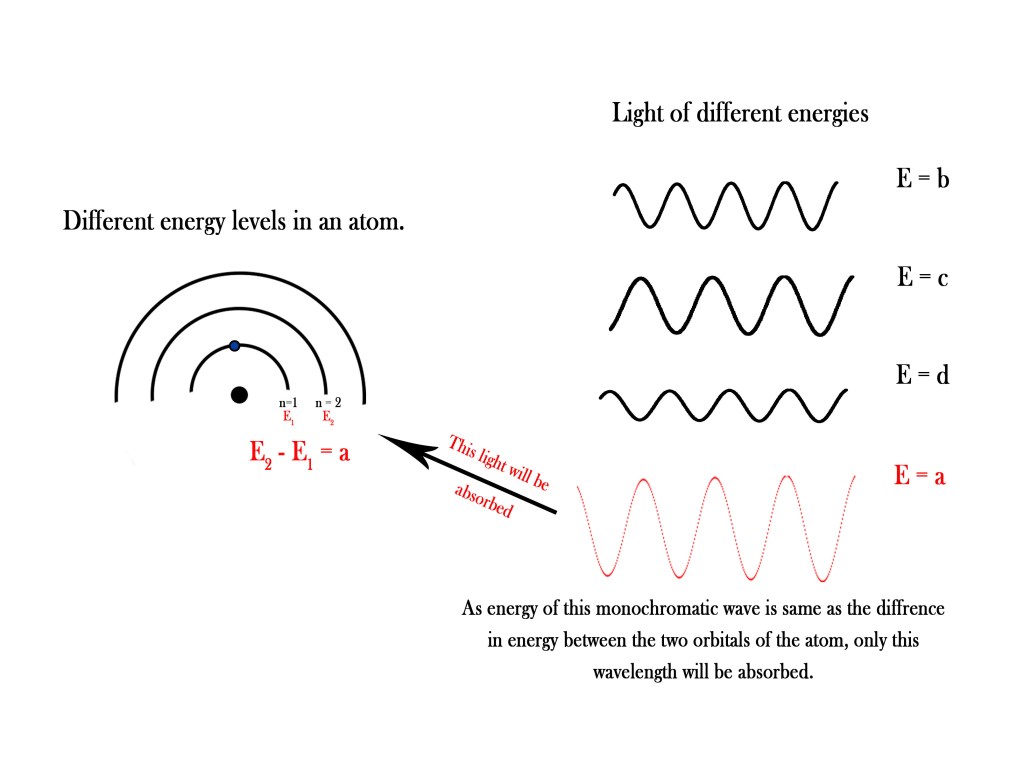
As seen in the above figure, the energy of the first orbital n=1 is E1 and that of the second orbital of the atom n=2 is E2. Let us say that the difference in these energies is ‘a. On the right hand are monochromatic waves of different wavelengths. Each wave has different energy (a,b,c,d). Only the fourth wave has exact energy equivalent to the energy difference between two orbitals and thus only the light with E = a will be absorbed by the electron in the first orbital of this atom. The electron (in n=1) will absorb this energy and get excited to n=2.
Lambert-Beer’s law
This law gives us the probability or rate of absorption of light by a medium. Actually, there are two laws – Lambert’s law and Beer’s law. Together they are referred to as Lambert-Beer’s law.
When a ray of monochromatic light passes through an absorbing medium, its intensity decreases exponentially as the length of the absorbing medium/thickness of the layer increases.
Lambert’s law.
Thus, the loss of light intensity, when it propagates in a medium, is directly proportional to its path length.
What does this mean?
(Monochromatic means – MONO = one , CHROMA = light .. Light of a specific wavelength)
As seen in the above figure, green laser light is made to pass through a colored solution in a measuring cylinder. As the light passes through this solution, its intensity decreases – on the top the light is bright . However, it fades as it passes through the cylinder. This is because the light is absorbed by the solution. Thus, the intensity of transmitted light is very less compared to the incident light.

Beer’s Law
Much later in 1852, August Beer discovered another relation between the concentration of the solution and the absorption of light.
We shall study this law in the next post. Till then,
Be a perpetual student of life and keep learning…
References and further reading –
1. Physical Chemistry by W J Moore (First edition)
2.https://science.jrank.org/pages/5151/Photochemistry-basic-laws-photochemistry.html
