This post is dedicated to understanding the empirical formula concept in organic chemistry. In the successive posts, we shall study other types of formulae too. However, it is imperative that one understands the empirical formula concept correctly, as it forms the basis of understanding the structures of organic molecules.
The Empirical Formula
The empirical formula is the formula, showing the simplest whole number ratio of atoms of different elements in a molecule.
e.g. Benzene’s empirical formula is CH. This formula shows that, in a benzene molecule, the ratio of carbon to hydrogen atoms is 1:1 (# of C-atoms = # of H-atoms).

But finding these ratios is not always easy. Let us try to understand the mathematical and chemical aspects of this term.
So what does the simplest whole number ratio mean?
As we have already studied in mathematics, a whole number means a number without a decimal point or without any fractions. 0,1,2,3,4,5….etc, are all whole numbers.
In a benzene molecule, there are six carbon atoms and fourteen hydrogen atoms.

Now, consider a molecule, C6H14 – Hexane. The ratio of no.of carbon atoms to hydrogen atoms in this molecule is –

Undoubtedly, ‘6/14’ is a whole number ratio, as both 6 and 14 are whole numbers. However, this is NOT the simplest whole-number ratio. Mathematically, we can simplify this fraction further by dividing both the numerator and denominator by 2 to yield 3/7.
However, if we further try to simplify this fraction, we get a decimal figure. Thus, 3/7 is the simplest whole-number ratio for C6H14. Thus, the empirical formula for hexane is C3H7.
Why do we need whole numbers only?
It is because we are talking about atoms. The number of atoms in a molecule is always a whole number. We cannot have 2.33 atoms of hydrogen participating in a reaction. Atom as an entity is a whole and cannot be subdivided in molecule formation. Thus, the number of atoms is always a whole number.
Procedure to calculate empirical formula (3 steps)-
Step 1 – Find the mass of each element in the sample.
i) Generally, the percentage composition (% composition) of the elements in a compound is given. The % composition refers to the amount/mass of that element in the given sample. If we assume the sample to be 100 g, the % composition is equal to the mass of the element in 100 g of the sample.
e.g.– In the above example (benzene,C6H6) , C= 50% and H= 50%.
If we take 100g of benzene as the sample,
50% of benzene = 50g will be carbon and
50% of benzene =50g will be hydrogen.
However, if the sample is not precisely 100g, then the % composition is NOT equal to the mass of the element.
Thus, if we have 160g of benzene as the sample, the mass of C and H present in it will be –
50% of 160g of benzene = 80g of Carbon and
50% of 160g of benzene = 80g of Hydrogen.
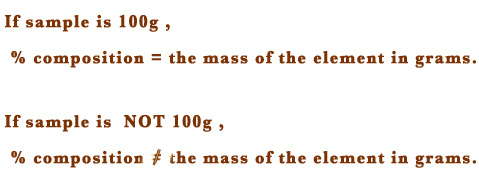
ii) If the sum of masses of the given element does not add up to 100, it indicates that oxygen is present.
e.g.- If we are asked to calculate the empirical formula of a substance that contains 40% carbon and 6.66% hydrogen, we should immediately conclude that oxygen is present.
How much oxygen is present?
40+6.66 = 46.66% of Carbon and hydrogen in 100 g of sample.
∴ amount of oxygen = 100 – 46.66 = 53.34%.
Step 2 – Converting atomic mass to moles.
For most stoichiometric calculations (calculations involving measurement of weight/amount of substances), we work with the concept of moles. To convert the amount of a substance from grams to moles, we just have to divide the amount in grams by the atomic weight of that element (the atomic weight is obtained from the periodic table). The quantity thus obtained gives no moles of that element present.

Note that, an empirical formula just gives the ratio of no.of atoms of the elements present in the molecule. In numerical problems, we are given the %composition of these elements. The %composition refers to the mass of the element. These two are different quantities.
e.g. The empirical formula of hexane is C3H7. This means that for every 3 atoms of carbon, there will be 7 atoms of hydrogen. However, it DOES NOT imply that FOR EVERY 3 gms OF CARBON THERE WILL BE 7 grams OF HYDROGEN.
Also, 1mole of any substance = 6.023 × 1023 atoms.
Thus, we need the mole concept to convert the mass ratios into atom ratios!
Step 3 – Find the simplest ratio and round it off to get a whole number.
In step 2, we obtain the number of moles of each element present in the compound. However, for empirical formula consideration, we need to determine the simplest whole-number ratio of the moles.
How to find the simplest ratio of moles?
Find the lowest number of moles quantity among all elements present in the compound. Divide all the ‘# of moles’ quantities by this number and then round off the figures to obtain simple whole numbers. The rounding off is done using the rules for rounding off significant numbers. (Check out the example below to understand the procedure correctly).
Lets us solve some examples to figure out how to do the calculations.
Problem – The percentage composition of an aromatic hydrocarbon is – C= 92.4% and H= 7.6%. Find the empirical formula of this compound.
Solution – We find the solution in a tabular form as it makes things look easy. Here, the amount of sample is not given, so we assume it to be 100g.
|
Element |
% composition (Given) |
Weight in 100g |
Atomic weight (At.wt) (from PT) |
# moles = (W/ At.wt) |
Simplest ratio(SR)= (# moles/lowest quotient) | Rounding off to a Whole Number. |
|
C |
92.4% |
92.4g |
12 |
(92.4/12) =7.7 |
(7.7/7.6) = 1.01 |
1 |
|
H |
7.6% |
7.6g |
1 |
(7.6/1) = 7.6 |
(7.6/7.6)= 1 |
1 |
∴ The empirical formula is CH.
Step 1– In this example, the %composition is given. As 92.4 + 7.6 = 100, we can conclude that this compound does not contain oxygen. It only has carbon and hydrogen elements in it.
Step 2 – We find out the atomic weight of each element from the periodic table. Divide the mass in grams with this atomic weight to obtain the number of moles. As seen above, we have 7.7 moles of carbon and 7.6 moles of hydrogen in this compound.
Step 3– The lowest number of moles quantity is 7.6 moles. We divide the number of moles by this number 7.6. For carbon, we obtain an answer equal to 1.01. We round off this number to the nearest whole number,1.
In the next post, we shall study how to calculate molecular formulas from empirical formulas and solve many more problems to deepen our understanding of these concepts. Till then,
Be a perpetual student of life and keep learning…
Good day!
References and further reading –
1.http://www.chem.tamu.edu/class/fyp/stone/tutorialnotefiles/empirical.htm
2.http://www.dummies.com/education/science/chemistry/how-to-calculate-the-empirical-formula-of-a-compound/