This post is all about asking the right questions to arrive at the desired answer. We will study certain observations that do not follow the expected path and ask some important questions. These questions will eventually lead us to the answer in the form of a rule – the DRAGO RULE.
This rule was stated to explain certain observations, which could not be explained by hybridization theory alone.
Why is the Drago rule needed?
1] To explain anomalous observations in bond angles of Group 15 and 16 hydrides-
Group 15 elements → Nitrogen (N), Phosphorous (P), Arsenic (As), and Antimony (Sb).
Hydrides of these elements → Ammonia (NH3), Phosphine (PH3), Arsine (ArH3), Stibine (SbH3).
The bond angles for the above four compounds are –
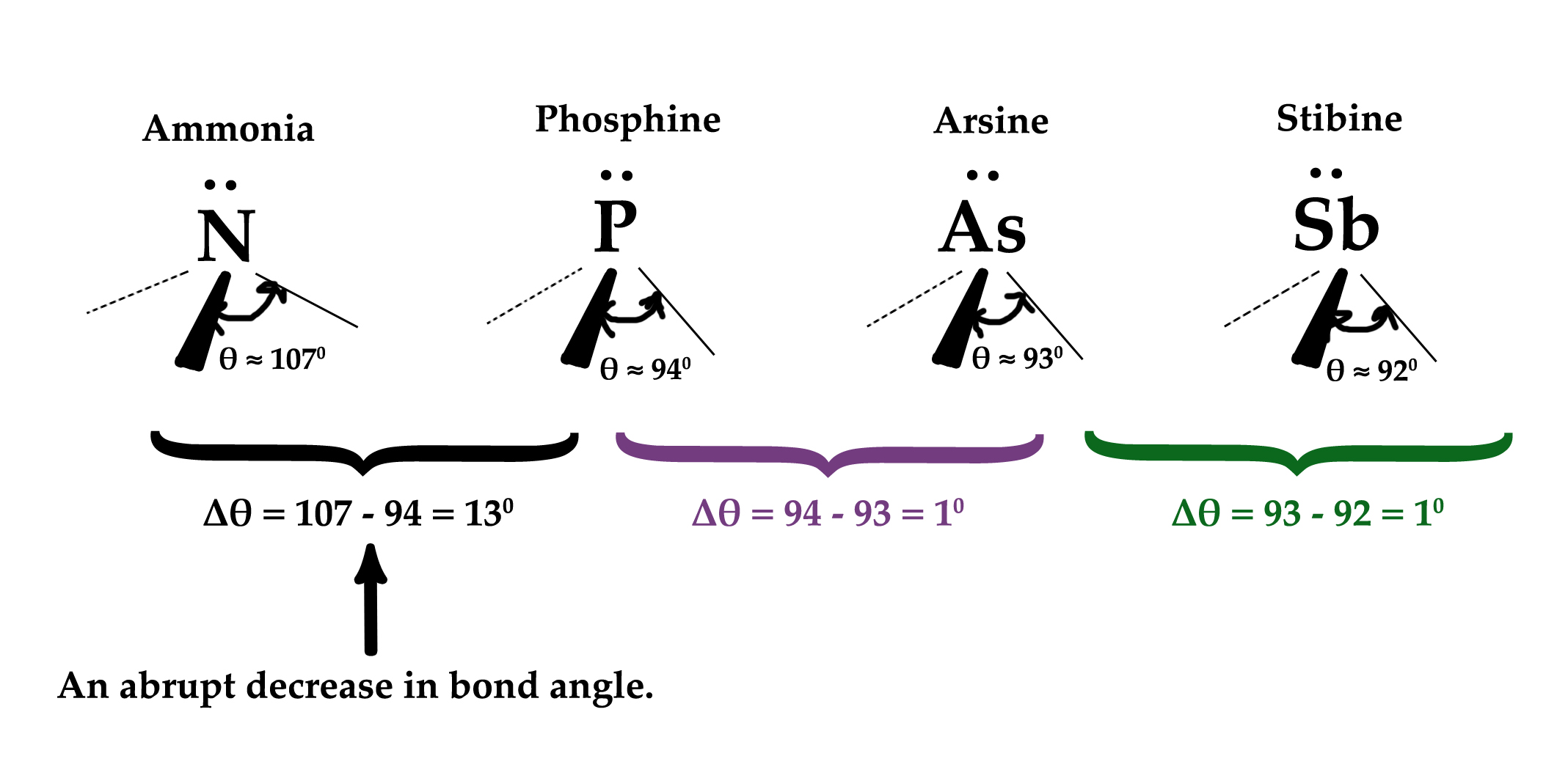
As seen in the above figure, the bond angle drops suddenly from ammonia to phosphine by 13º. There is just a 1º difference between bond angles of phosphine, arsine, and stibine. Why does the bond angle drop so much going from ammonia to phosphine?
A similar observation can be made for group 16 elements too.
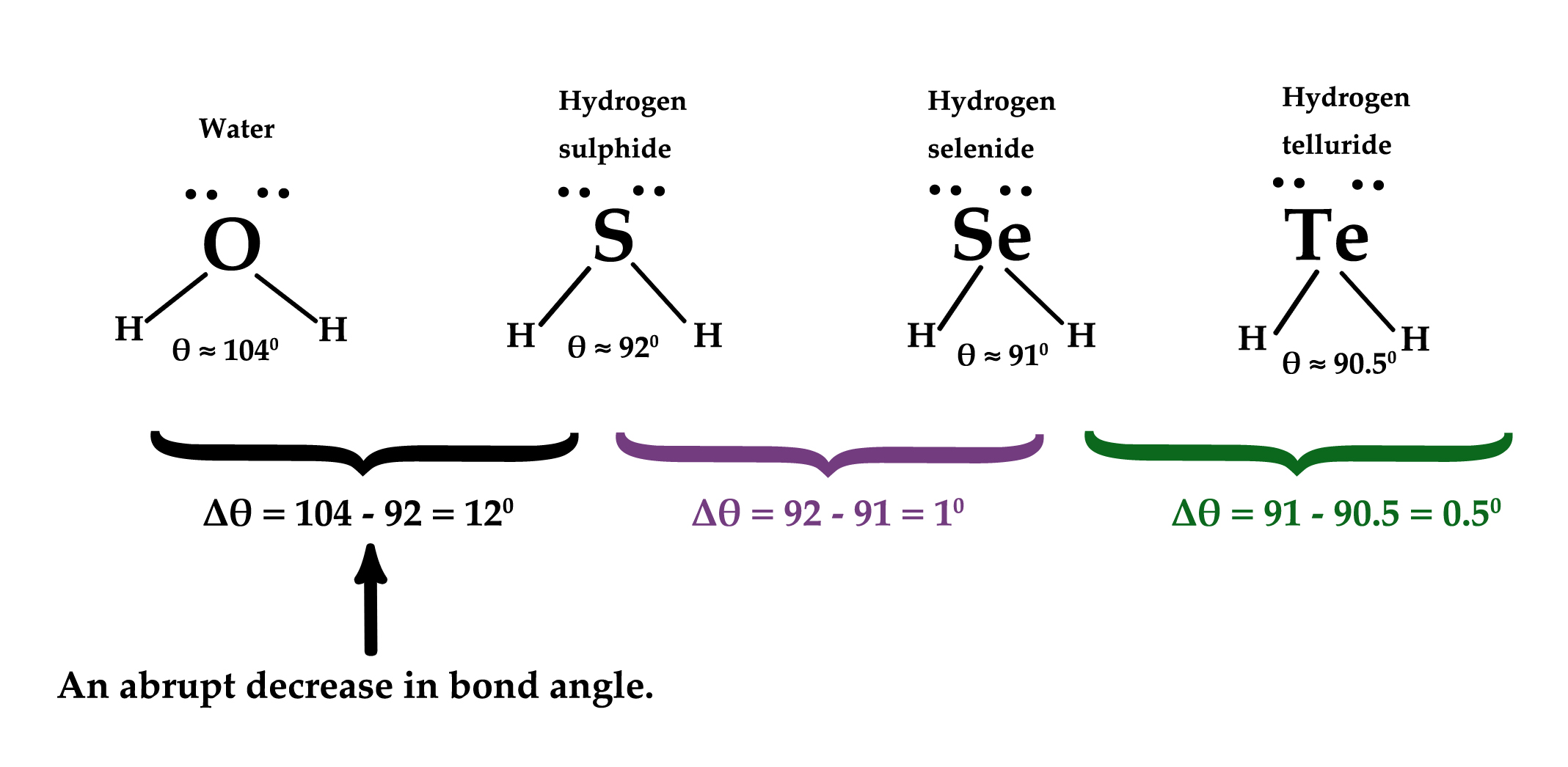
Oxygen, sulfur, selenium, and tellurium are all Group 16 elements. Their hydrides also show behavior as Group 15 elements. Why does the bond angle drop so much going from water to H2S?
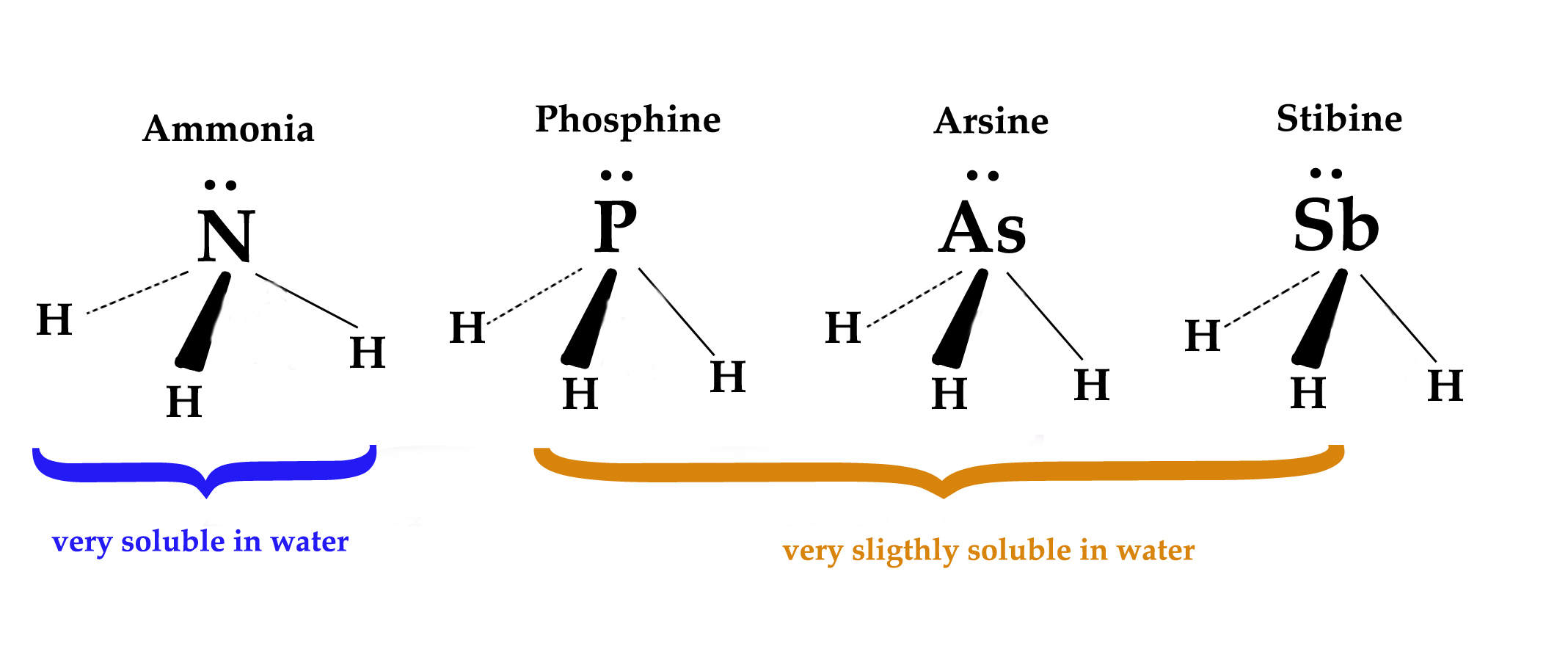
2] To explain solubility differences of certain compounds –
In the above set of compounds, it is very surprising to see that ammonia is very easily soluble in water. It forms ammonium hydroxide (NH4OH)- which we all have extensively used in our laboratory. However, phosphine, arsine, and stibine do not dissolve /partially dissolve in water. If their outer electronic configuration and the hybridization they undergo are the same, then all these compounds should exhibit similar solubility. However, practical observations tell a different story! What is the reason for this solubility difference?
3] To explain the reaction of ammonia and phosphine with acids-
Ammonia readily forms ammonium chloride with dilute hydrochloric acid (HCl). However, phosphine only reacts with concentrated HCl. Why can’t phosphine react with the dilute acid?
NH3+ dil HCl → NH4Cl .
PH3+ dil HCl → NO REACTION.
PH3+ conc HCl → PH4Cl.
4] Water and ammonia act as ligands and form many coordination compounds. Why don’t other compounds like PH3, ArH3, H2Se never form such coordination compounds?
(We shall learn coordination chemistry in much detail in the chapters to follow. For now just remember that coordination chemistry deals with compounds having a central metal ion bonded to negative ions or neutral species, which are called ligands).
All the above discrepancies can be explained by Drago’s rule. In the next post, we shall study the rule and how it explains the odd behavior of the molecules. Till then,
Be a perpetual student of life and keep learning…
Good day!