THE DRAGO RULE.
Drago rule states that if –
- the central atom has at least one lone pair of electron on it.
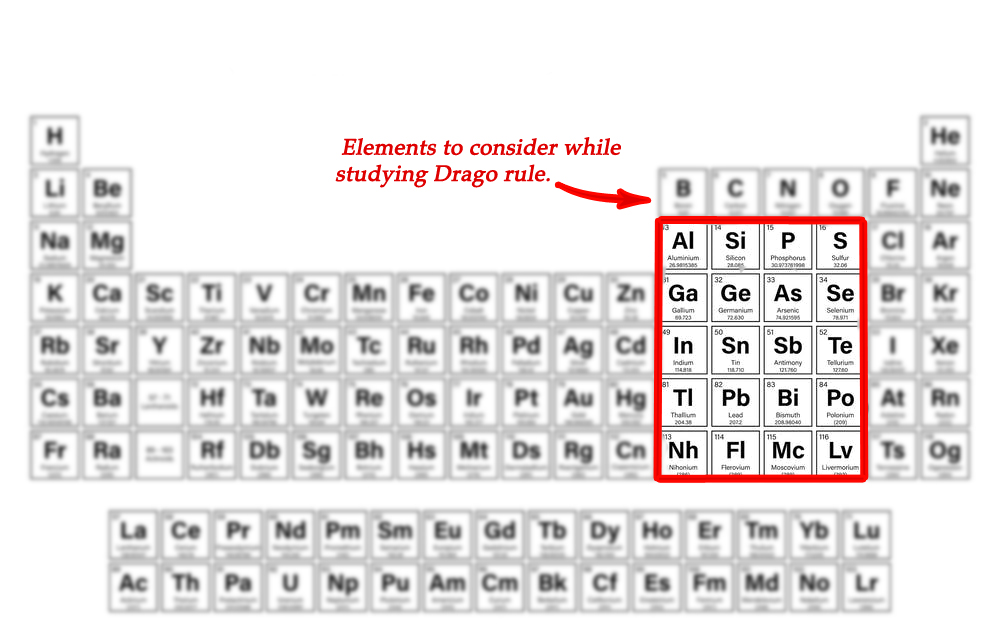
- the central atom belongs to group 13,14 ,15 or 16 and is from 3rd to 7th period.
- if electronegativity of central element is 2.5 or less
- no. of sigma bonds+ lone pair = 4
then, THERE IS NO NEED TO CONSIDER HYBRIDIZATION OF THAT ELEMENT.
Consider phosphine(PH3). The central atom in phosphine is phosphorous.
Phosphorous –
i) belongs to group 15 and is in the third row of the periodic table (see the table above).
ii) has an electronegativity value, χ = 2.19 and
iii) in phosphine molecule, it has a lone pair of electrons too.
So, according to Drago’s rule, hybridization does not take place. The atomic orbitals just overlap to form bonds.
If we closely look at the structure of Phosphine, we would know the above statement to be true.
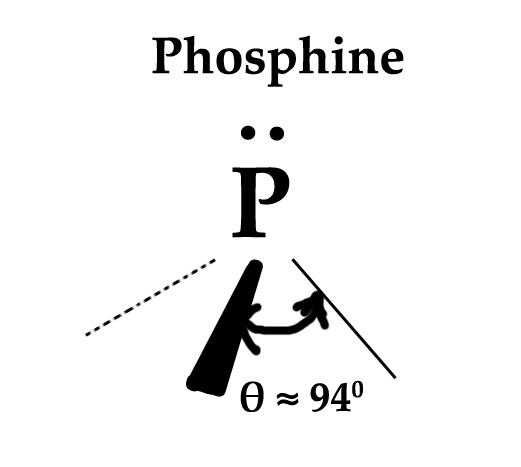
The bond angle in phosphine is 94º . If we apply the formula from post 69 ,
fs=cosθ/(cosθ-1)
∴fs= (-0.069) /( – 0.069-1)
∴fs= -0.069 / -1.069
∴fs= 0.064
%s character = fs×100 = 0.064 ×100 = 6.4 %
This means that % s character in the P- H bonds is just 6.4% ≈ 6%.
Thus, there is only a 6% s- character in P-H bonds. So, the s-character in three P-H bonds will be 6 × 3 = 18%. So, we can conclude that the lone pair of electrons are in orbitals which have (100 – 18 =) 82% s character! Now, no hybridized orbital ever has this high % of s- character! (The maximum %s character is 50% in sp orbital) .This means that the lone pair of electrons are in a pure s- orbital and NOT IN ANY HYBRIDIZED ORBITAL.
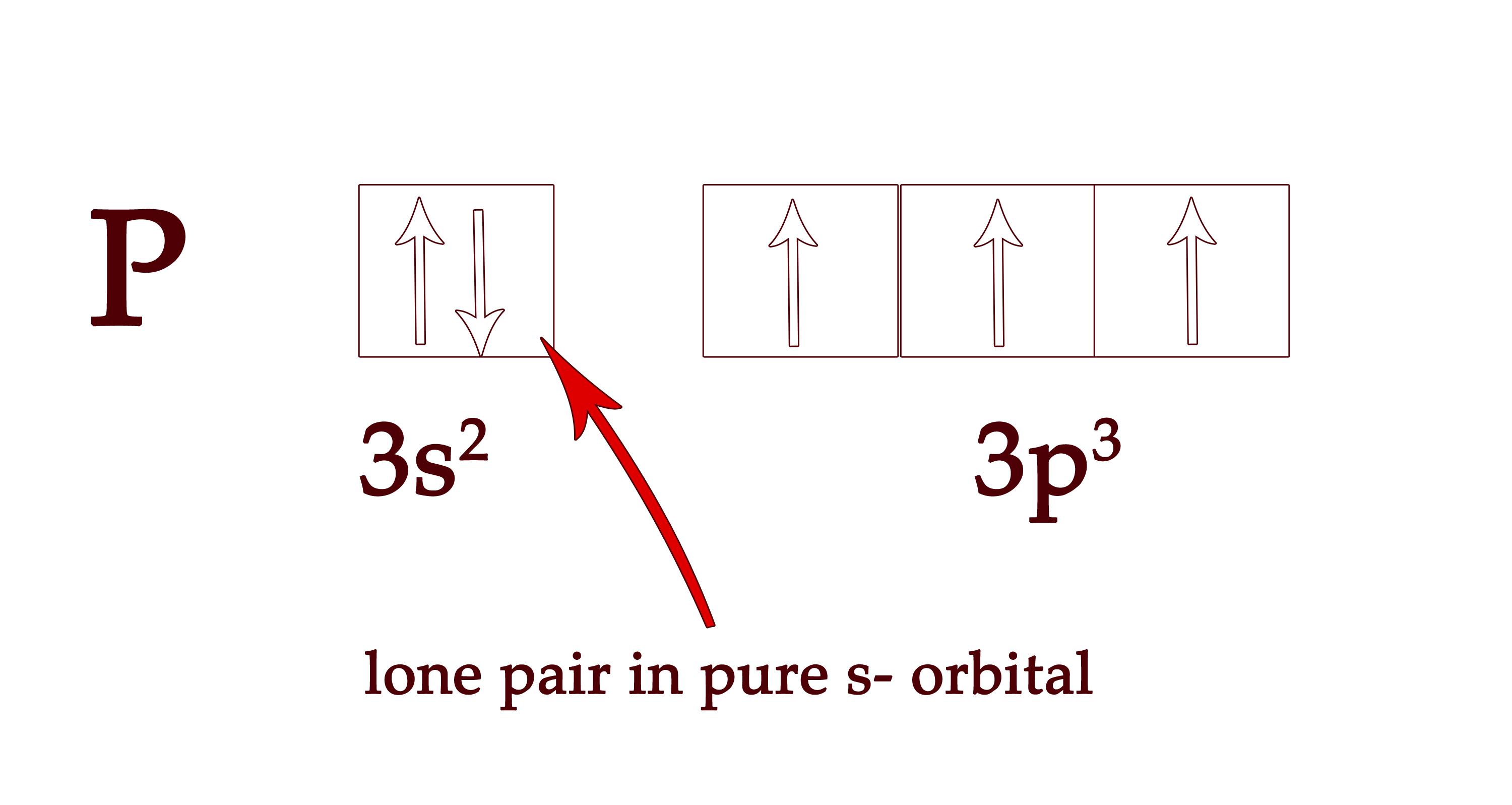
The same logic holds true for all the other elements too. Now that we have established that the lone pair is NOT in any hybrid orbital but in a s- orbital, we can explain all the observations we listed in the earlier post.
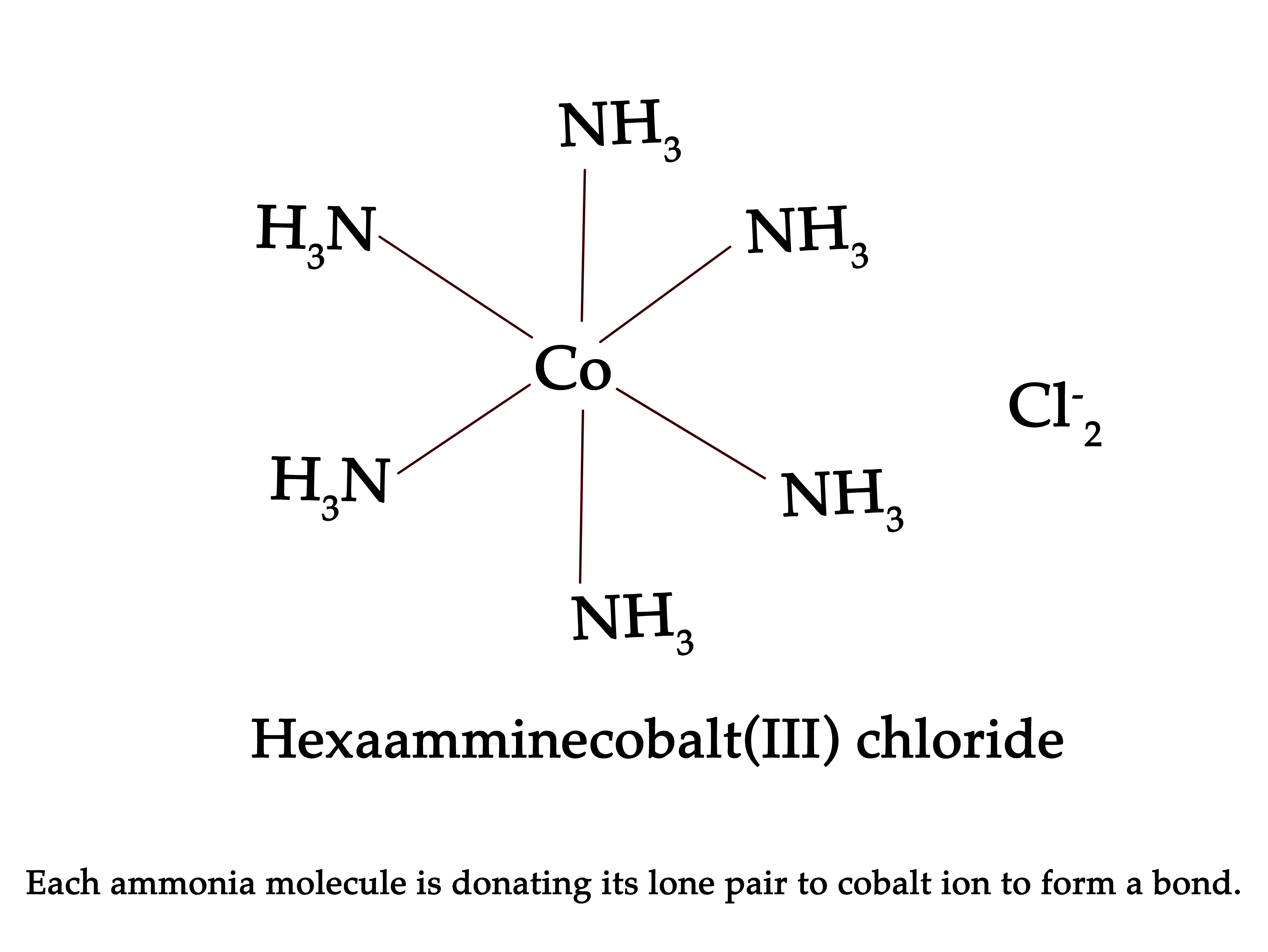
As the lone pair is in a s- orbital, it is closer to the nucleus and so very tightly bound to it. Thus, this lone pair of electrons is NOT available for sharing or donating. So, it takes concentrated acid to react with phosphine. Also, as these lone pairs cannot be shared, these compounds do not form coordination compounds. Ammonia on the other hand can lend its lone pair to form coordinate covalent bonds and so can form coordination compounds.
e.g.- Ammonia dissolves in water as it forms hydrogen bonds with water molecules. This is possible as the lone pair of electrons are available on nitrogen.
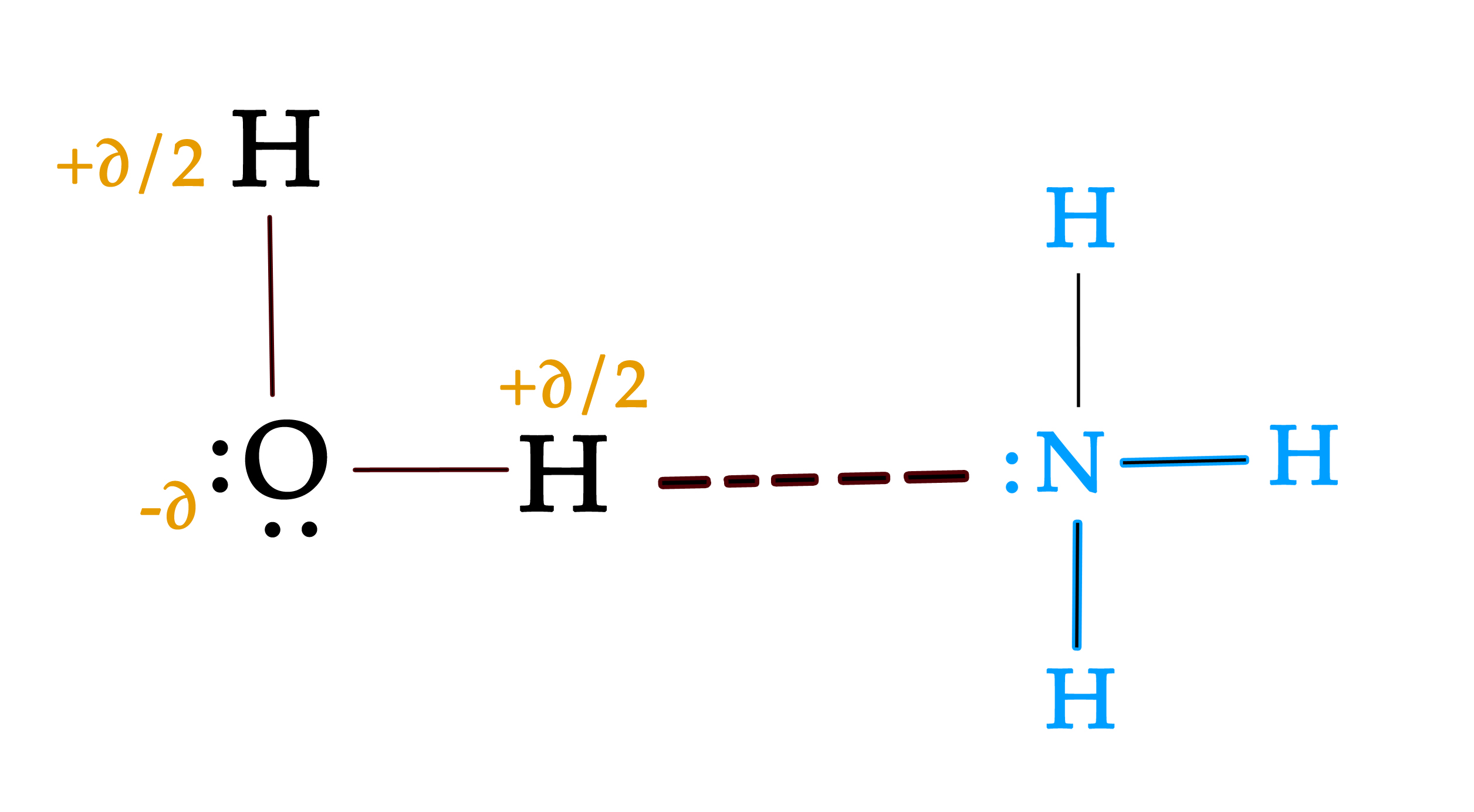
All the questions which we considered in the last post, can be answered by Drago’s rule. The simple explanation to those questions is, the elements of group 13-16 and 3rd- 7th period do not have a lone pair in any hybridized orbital. This lone pair is thus unavailable.
As opposed to this, hybridization takes place in the elements of the 2nd period. Thus, the lone pairs occupy the hybrid orbitals, which have less %s character. Naturally, they are less tightly bound to the nucleus and are available for sharing or bonding.
In our next post, we shall try to solve some problems related to hybridization theory. Till then,
Be a perpetual student of life and keep learning…
Good Day!
References and Further Reading –
1.https://www.embibe.com/ask/question/what-is-dragons-rule-865
2.https://www.quora.com/What-is-drago-rule
Image source –
Thank you ma’am ❤️👍
LikeLike
My pleasure 🙂
LikeLike
Tysm Mam! ❤
LikeLike
My pleasure !
LikeLike
It’s really helpful for me.
LikeLike
I am glad I could help 🙂
LikeLike
Really very helpful ma’am .., thank you ma’am
LikeLike
Ma’am though no hybridised orbital has such high percentage s character what does it mean when you say that the percentage s character is 82 percent.
LikeLike
One s- orbital is used to make a hybrid orbital – that means 100% of that orbital is used to create something new. In this case , the P-H bonds have only 6% s-character in them. There are three P-H bonds , so 3* 6= 18% s character in these bonds.
Which means the rest 100- 18= 82% is left. No hybrid orbital can have such a high value of s-character in it. Thus, we conclude that the orbital containing the lone pair of electrons is NOT a hybrid orbital. It is a pure s- orbital.
Hope this helps 🙂
LikeLike
Ma’am though no hybridised orbital has a percentage s character as high, what exactly does it mean when you say that the percentage s character of is 82 percent. And also if a single P-H bond has a percentage s character of 6 percent and all three bonds collectively have a percentage s character of 18 percent, why is it that this value was subtracted from 100 to obtain 82 percentage as the answer. I don’t understand, could you please help.
LikeLike
Hi Bruno.. I have calculated the %s- character for all the three bonds in phosphine using the Draco rule. The value of s- character in each bond comes to 6%. Thus, for 3 P-H bonds the value will be 3*6 = 18 %.
As you know, the entire s-orbital (100%) of phosphorous atom is used to make phosphine. So if 18% has gone in making the phosphine bonds, the remaining s- orbital (100 – 18 = 82%) must have gone in an orbital containing the lone pairs. So, the 3s2 orbital , which contains the lone pair has 82% s- character.
However, we know that only three types of hybridization are possible –
1) sp3 hybrid orbital – 25% s- character
2)sp2 hybrid orbital- 33% s- character
3)sp hybrid orbital – 50% s- character.
The s- character in 3s2 orbital is much more than 50%.
So , if the s-character is more , we can safely conclude that the orbital cannot be a hybrid one.
In the case of phosphine, the lone pairs are in an orbital with 82% s- character. Thus, the orbital holding these lone pair of electrons cannot be a hybrid orbital. It HAS TO BE a regular pure s- orbital.
Hope this helps .. feel free to get back if you have further queries.
LikeLike
Thank you so much ma’am. This definitely helped 😁
LikeLike
My pleasure 🙂
LikeLike