We continue to study the physical manifestations of H- bonds and their importance.
Protein folding
Hydrogen bonds are an integral part of the protein structure. We will study the protein structure in detail in later posts. Here, we just need to know that proteins are made of amino acids(aa). As the name suggests, amino acids have both amino (-NH2) and carboxylic acid (-COOH) groups in their structure.

One amino acid gets attached to another amino acid by a peptide bond as shown in the figure below –
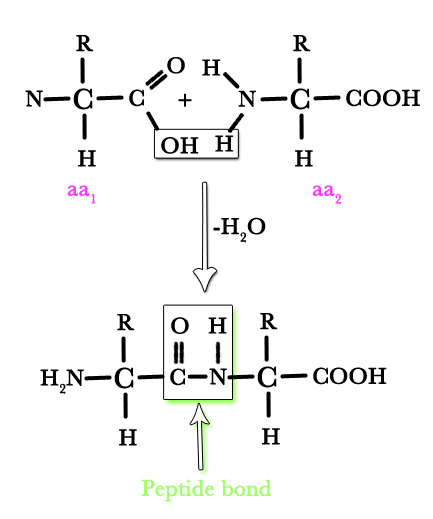
Many amino acids come together to form a polypeptide. This is the primary structure of a protein.

The secondary structure of proteins has hydrogen bonds in them. The secondary structure is formed by folding the primary structure into an alpha(α)-helix or beta(β)- pleated sheet.

This video will give a good idea of how protein structure is –
Only this secondary structure with hydrogen bonds is functional. So, the proteins in our body have this secondary structure and thus, it is the hydrogen bonds that are responsible for making them functional !! Proteins are found in every cell of our body. Hairs, Nails, enzymes, blood, hormones, antibodies, DNA are all made of proteins. So, now we understand the importance of hydrogen bonds and their role in forming the secondary structure of proteins.
In the next post, we will solve some problems related to chemical bonding before winding up the discussion on chemical bonding. Till then,
Be a perpetual student of life and keep learning ….
Good Day!
Thanks for this
LikeLike