From this post onwards we begin discussing a new topic in organic chemistry. This topic deals with a certain class of organic compounds, which are called aromatic. What are these aromatic compounds? Why are they so important? What exactly does aromaticity mean? What distinguishes this class of compounds from others? What kind of reactions are the characteristics of these compounds? Let us learn all about this phenomenon – AROMATICITY.
In the previous posts, we studied that benzene molecule exhibits exceptional stability. This is because benzene is aromatic!
Aromatic compounds like benzene, toluene, xylene, etc find application in the production of a wide variety of day-to-day materials. About 35 million tons of aromatic compounds are produced every year to manufacture various materials like medicines(drugs), dyes, pesticides, cosmetics, polymers, explosives, etc. The human body also contains aromatic compounds like tryptophan and phenylalanine, which are amino acids.
AROMATICITY.
The term aromatic is derived from the word ‘aroma‘, which means fragrance. Most of the aromatic compounds have a peculiar aroma, thus they are referred to as ‘aromatic’.
Aromatic compounds have conjugated/alternate double bonds in their structure.

There is resonance between these conjugated bonds and thus the molecule has a delocalized π electron cloud. These compounds have extra stability owing to their structure. This special stability is called aromaticity.
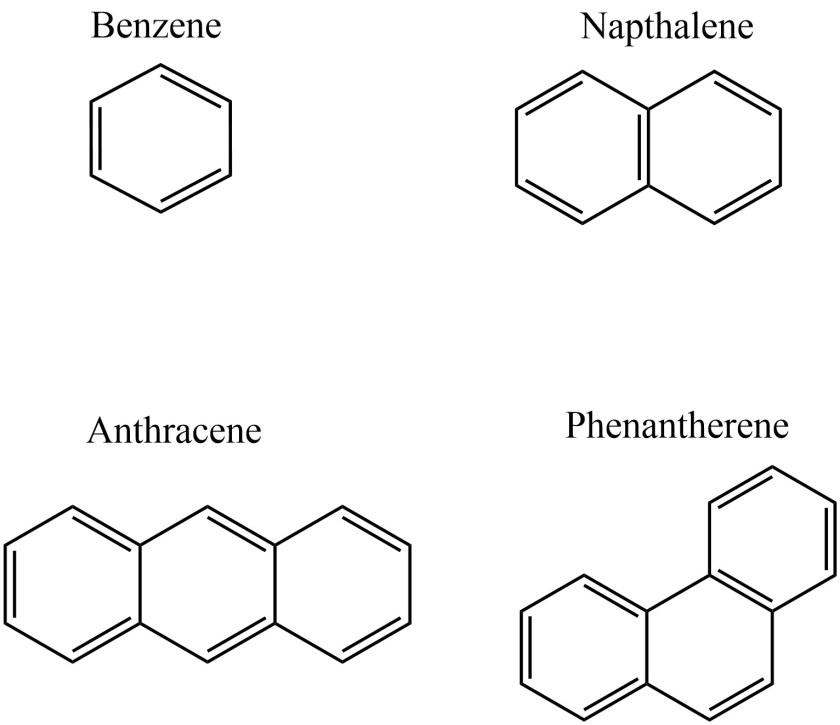
e.g. – Benzene, Naphthalene, Anthracene, Phenanthrene,etc.
Aromaticity is the ability of many cyclic compounds containing conjugation in their ring, to –
i) undergo substitution reactions rather than addition reactions
ii)to resist oxidation and
iii) to exhibit marked stability of their rings.
e.g.- When bromine is added to benzene, it DOES NOT ADD across the double bond as it adds across an alkene. Instead, we get a substitution reaction as shown below –

We shall learn aromaticity in great detail in the upcoming posts. In the next post, we will start discussing how aromaticity is described in MO terminology. Till then,
Be a perpetual student of life and keep learning…
Good Day!
Image source –