From this post onwards we will start getting acquainted with some fundamentals of analytical chemistry. What is analytical chemistry? What all topics does it encompass? How useful is it ? We will answer all the above questions and learn some basic terminologies used in analytical chemistry.
What is analytical chemistry?
As the name suggests , analytical chemistry deals with analysis. It is imperative in many areas of our modern life to analyse different things – where quality of substances needs to be assessed.
Analytical chemistry is the science of obtaining, identifying, processing and communicating information about the composition and structure of matter.
This branch of chemistry provides answers to questions like –
What is present? – Qualitative Analysis
How much is present? – Quantitative Analysis
Why should we analyze things?
Think about all the times you have wanted to know the nutritional contents of your packed food items. Does the food contain trans fat? If yes , then how much ? While taking your medication, your doctor prescribes a dosage for various drugs. The dosage of medication is very critical in most cases. Forensic science studies blood , DNA samples to solve criminal cases.The pathological tests like checking your blood samples, urine sample, body fluids also use analytical chemistry techniques.
Analytical data is used in a wide range of disciplines like –
1)Quality Control – In industries there are stringent requirements approved by legislation for the composition of the components manufactured. The level of impurities/contaminants need to be checked to ensure best quality products. The food, pharmaceutical, electrical goods, horticulture ,water industries , geological studies are among the many industries that need to follow strict rules regarding the composition of the manufactured product. Analytical chemistry plays a crucial role in quality control in these industries.
2)Monitoring and control of pollutants – Water, air pollution studies use analytical techniques to determine the level of toxic materials in water and air. Analytical chemists also provide remedial solution in such matters. e.g.– Waste management methods.
3)Clinical studies – Various pathological tests carried out for patients are a part of analytical chemistry study. e.g.- MRI scans, blood analysis, urine analysis etc.
Analytical chemistry finds applications in various other fields like analyzing samples from ores, space, toxicology studies etc. In short analytical chemistry is used to study anything and everything under the sun!
A typical example of the importance of analytical chemistry can be seen in the film Erin Brockovich.

This film narrates a true story of a woman, who finds out how the groundwater is contaminated with hexavalent chromium. The hexavalent chromium was the cause of many medical problems of the residents of that region.It is only through various analytical methods that such issues can be studied.
Basic steps in analytical chemistry.
Analytical chemistry deals with the following five basic steps –
1.Identification and defining the problem and to find out what type of measurement is required. Do we need to find out what is present or we need to find out how much is present or both?
2. Design the experimental procedure to solve the problem. This step involves finding out how to get a correct sample , which experimental method to consider etc.
3. Conducting the experiment, and gather data about the analyte.
4. Analyze the experimental data.
5. Propose a solution to the problem.
Common terms involved in analytical chemistry.
Before diving deeper into the study of analytical techniques, it is crucial that we know the various terms used by analytical chemists. The basic terms used are –
- Analysis – Analysis provides chemical and physical information about the sample. We study the identity , concentration and various properties of the sample during analysis
- Sample(noun) – The specimen to be analysed is called sample. Sample consist of two parts namely –
i) Analyte – The components of interest in a sample upon which analysis needs to be performed is called the analyte.
ii)Matrix – The remainder of the sample, without the analyte , is the matrix. The analyte is sort of dispersed in matrix.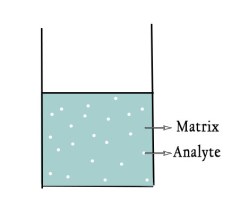
- Sample (verb)– To collect one or more samples.
- Test portion – A portion of the collected sample that is processed and measured.
- Qualitative analysis – Analysis done to determine the identity, structure and physical properties of a substance. e.g. – Spectroscopic methods.
- Quantitative analysis– Analysis done to determine the amount of analyte in the sample. e.g.– Gravimetric analysis.
- Detector – Device used to analyze the analyte in the sample. e.g.- An electrometer is used to determine electrical conductivity of the analyte in the sample.
- Signal – The result or output shown by the detector.
- Interference – These are substances which have similar properties as the analyte. Thus, they interfere with the measurement/detection of the analyte.
- Accuracy and Precision – See post 16
We will continue to learn some more terms in the next post. Till then,
Be a perpetual student of life and keep learning….
Good day !
Image source-