In this post, we will start discussing the second law of photochemistry, namely the STARK -EINSTEIN LAW.
STARK- EINSTEIN LAW
This law was formulated by Johannes Stark and Albert Einstein. This is law can be thought of as the application of quantum theory to photochemistry. The quantum theory deals at the level of sub-atomic particles and photons. Thus, this theory tries to explain what happens when a photon of light is absorbed at the sub atomic level.
This law states that –
“Each atom or molecule absorbs a single quantum of light (photon) in the process of getting excited.”

M + hν → M*
In the above expression , hν represents a single photon of monochromatic light (light of a particular wavelength).
Extending this law further, we can conclude that, if 1 atom/molecule absorbs only 1 photon , then, 1 mole of a substance will absorb 1 mole of photons while reacting. The energy associated with 1 mole of photons is termed as 1 Einstein of energy (E).

1 Einstein (E) is the amount of energy of 1 mole of photons of a given frequency absorbed by the system.
1E = NA hν= NA h c/λ, where
NA ⇒ 1 mole of a substance / Avagadro’s number (6.023 × 10 23 atoms/molecules)
h ⇒ Planck’s constant(6.6261×10−34 J⋅Hz−1)
c ⇒ velocity of light (3 ×108m/s)
λ ⇒ Wavelength of light in nm
Thus, 1E = (6.023 × 10 23 × 6.626×10−34 × 3 ×108 )/ λ
∴ 1E= 119666/ λ J mol-1
e.g.- If we have 1 mole of photons of wavelength 100nm, then , their energy will be 119666/100 = 119.6 J mol-1.
As is evident from the above equation, the energy of photons is dependent on the wavelength. The energy inversely varies with the wavelength. This means that photons with higher frequency have lower energy and vice versa.
e.g.- Photons in the UV range have lower wavelengths than photons in the IR range. So, the energy of UV photons will be more than that of IR photons.
One very important point concerning this law is that – THIS LAW IS APPLICABLE ONLY TO THE PRIMARY PHOTOCHEMICAL PROCESS(we will study the primary and secondary process in the upcoming posts).
QUANTUM YEILD / QUANTUM EFFICIENCY (Φ)
This term was enunciated by Albert Einstein. This is a very important term used in photochemistry which quantitatively tells us , how much product is formed by absorption of how many photons. In short this term defines the efficiency of the photochemical process.
Quantum yield is defined as “the number of molecules of the substance undergoing the photochemical change per quantum of radiation absorbed.”
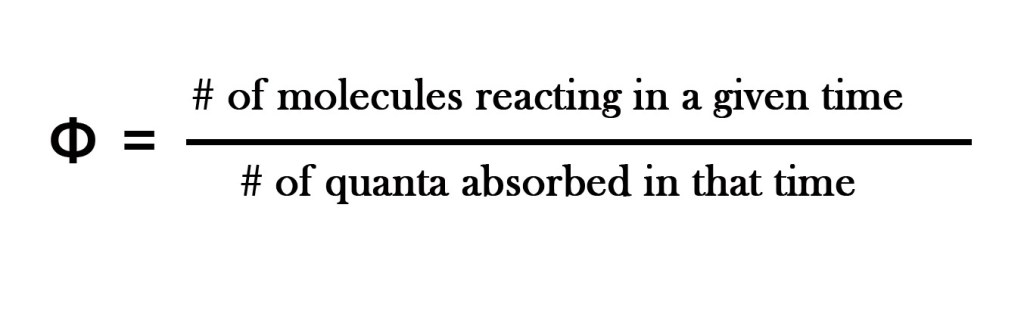
As photochemical reactions can be very complex. the quantum yield can vary from zero to a million.
For the primary process of a photochemical reaction, which obeys Stark – Einstein law , the quantum yield is one. This is because, one atom/molecule reacts by absorbing one photon of light. Thus, the quantum yield is one too. This means that, the reaction is absolutely efficient and all the light absorbed is effectively used to convert the reactants to another species.

We will talk more about this concept in the next post. Till then ,
Be a perpetual student of life and keep learning….
Good day !