The Molecular Orbital Theory (MOT).
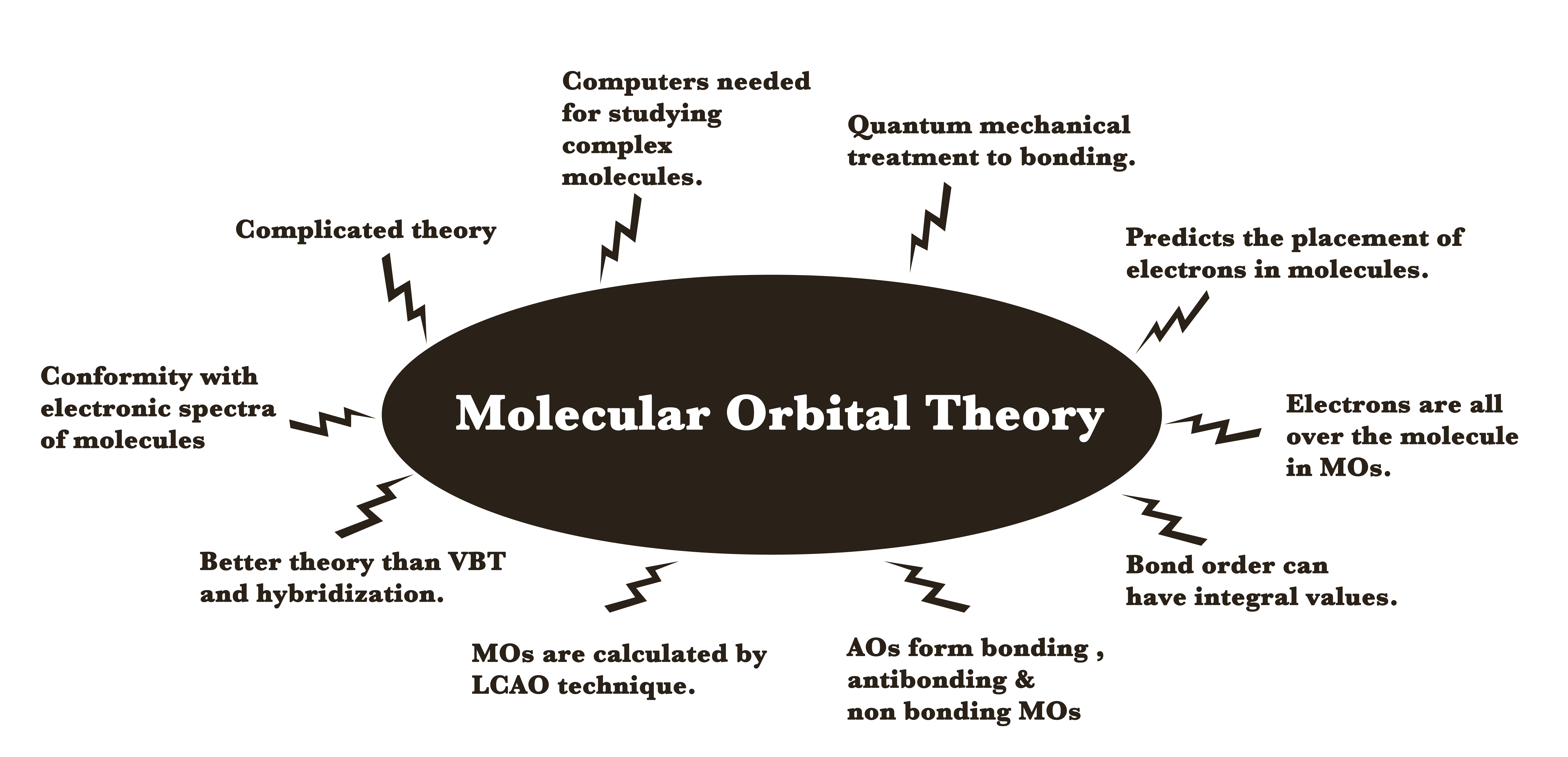
This theory assumes that the electrons, shared between the atoms, are spread throughout the molecule. These electrons are NOT localized on just one atom. Atomic orbitals overlap to form molecular orbitals(MOs) and the electrons occupy these MOs.
This theory is the quantum mechanical approach to bonding. The application of this theory involves complicated mathematics, especially for multi-atom molecules. Thus, computers are generally used while using MOT for complex species.
The scope of our syllabus encompasses the application of this theory to very simple diatomic molecules only. Learning this theory is important because this theory is very useful in predicting the structure, electronic spectra, and paramagnetism of molecules as compared to the VBT or hybridization theories.
Molecular orbital theory (MOT)
The postulates of this theory are –
- # of atomic orbitals combined = # of molecular orbitals formed.
- For every combination of the AOs a bonding molecular orbital (BMO) of lower energy and an anti bonding molecular orbital (ABMO) of higher energy is formed.
- Electrons will always to enter the orbital with lowest energy(Afbau principle).
- Electrons are delocalised i.e they are spread on all the atoms.
Similar to the hybridization theory, the MOT also considers that the no. of MOs formed are exactly equal to the no. of atomic orbitals(AOs) involved.
HYBRIDIZATION ⇒ # of AOs = # of hybrid orbitals.
MOT ⇒ # of AOs = # of MOs.
However, in MOT two kinds of MOs are formed namely –
1. BONDING MOLECULAR ORBITALS- BMO- (σ) and
2. ANTI BONDING MOLECULAR ORBITALS – ABMO – (σ*).
Before studying the BMOs and ABMOs, let us first note that, according to quantum mechanics, electrons behave both as particles and waves. This is called wave-particle duality. The orbitals are described by a wave function (ψ). To know more about a wavefunction read post 33.

In MOT, we model electrons as waves.
•The Bonding molecular orbitals (BMO) are formed when the electrons in AOs constructively interfere with each other. In this case, both waves are in phase.
• The Anti bonding molecular orbitals (ABMO) are formed when the electrons in AOs destructively interfere with each other i.e when the two waves are out of phase. Anti-bonding orbitals have node/s in them. A node is a point where the wave function is zero(ψ=0). At the node, the probability of finding electrons is zero.
The bonding and anti-bonding orbitals can be represented as shown in the figure below-
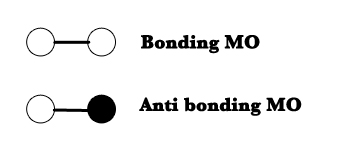
In bonding MOs, both the orbitals (circles in the figure above) are in the same phase. In an anti-bonding orbital, the electrons in the two orbitals are out of phase (the black circle indicates that it is not in phase with the white one).
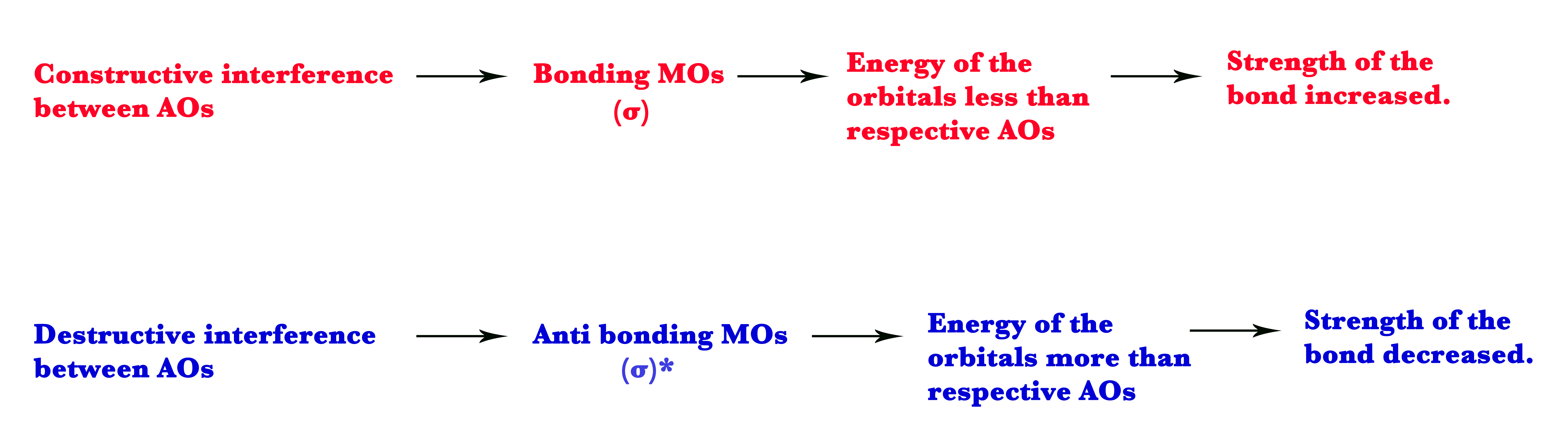

As seen in the MO diagram above, two atomic orbitals (AO) of atom 1 and atom 2 superpose to give two molecular orbitals (drawn between the grey dotted lines). One molecular orbital has energy lower than the two atomic orbitals. This is the bonding molecular orbital (BMO). The other orbital has energy more than the atomic orbitals. This is the anti-bonding molecular orbital (ABMO).
Electrons in bonding molecular orbitals make the bond stronger, as these orbitals are lower in energy than the AOs.On the other hand, electrons in anti-bonding molecular orbitals make the bond less stable, as these orbitals are higher in energy than the corresponding AOs.
•Non-bonding (NBMO) are formed when –
i) the electrons in AO do not have a similar energy to electrons in an orbital in another atom or
ii) the constructive interference with one lobe is canceled by destructive interference with another lobe of that orbital.
These orbitals have the same energy as the AOs.
For the overlap between the AOs to take place, the following conditions must be met –
- The AOs must have same symmetry.
- The AOs must have similar energy.
- The AOs must have spatial overlap i.e the atoms should be aligned correctly in space and must be close to each other for the overlap to take place.
BOND ORDER.
In MOT, we do not consider single, double, or triple bonds. We refer to these in terms of the bond order. In simple terms, bond order is the number of bonds between two atoms. The bond order is the measure of the stability of the bond. We will extensively talk about bond order in a later chapter. For now, we shall just learn to calculate bond order w.r.t MOT.

Stability of Molecules
The stability of a molecule can be determined by the following factors –
A] Stability based on bond order – In molecules, the bond order is a measure of the stability of the molecule. The higher the bond order, the higher is the stability.
↑ Bond Order ↑ Stability
e.g. – The bond order for nitrogen is 3 and that for oxygen is 2. So, nitrogen is a more stable molecule than oxygen.
Bond dissociation energy for N3 molecule = 941 kJ/mol.
Bond dissociation energy for 02 molecule = 425.9 kJ/mol.
Thus, it will take more energy to break a nitrogen molecule than an oxygen molecule as nitrogen is far more stable than oxygen.
B] Stability based on # of electrons in BMO and ABMO –
If # of electrons in BMO = NB
# of electrons in ABMO = NA
then,
i) If NB >NA → The molecule will be more stable as the net force of attraction exceeds the force of repulsion.
ii)If NA > NB→ The molecule will be unstable as the net force of repulsion will be more than the force of attraction.
iii) If NA = NB → The molecule will be unstable as the influence of electrons in the ABMO is more than those in BMO.
In the next few posts, we will continue talking about this theory. We shall see how orbitals interfere, how to draw MOT diagrams, and how to write configurations w.r.t this theory. Till then,
Be a perpetual student of life and keep learning…
Good Day!
References and Further Reading –